Learn how pulse chemisorption measures metal dispersion and surface area in supported catalysts, with advantages, limitations, and real published results.
Laboratories characterizing supported metal catalysts routinely face a method selection question that has no universally obvious answer. Volumetric chemisorption is the established reference technique, with decades of literature and standardized protocols behind it. Dynamic methods, specifically pulse chemisorption and temperature-programmed desorption (TPD), are faster and simpler. The practical question is whether the faster methods produce results that are consistent enough with volumetric data to be trusted for the same decisions.
Dynamic chemisorption refers to flow-based chemisorption techniques in which a reactive gas is passed over the catalyst in a carrier stream and the outlet composition is monitored continuously by a detector such as a thermal conductivity detector (TCD). The two most common dynamic methods are pulse chemisorption, which injects discrete gas pulses into the carrier stream, and temperature-programmed desorption (TPD), which monitors desorption of a pre-adsorbed gas during a controlled temperature ramp.
Volumetric chemisorption, also called static chemisorption, measures the quantity of gas adsorbed at a series of equilibrium pressures across a defined pressure range. The sample is exposed to the adsorbate in a closed system, and the amount adsorbed is calculated from the pressure change after equilibration at each point. This approach captures both strongly and weakly held adsorbate species and is considered the reference method for metal surface area measurement.
The key practical differences between these approaches are:
Two supported platinum catalysts with low metal loadings were selected for this comparison:
Carbon monoxide was chosen as the adsorbate because CO adsorption on platinum is well-established as a rapid process, making it well-suited for pulse chemisorption where fast kinetics are a requirement. Hydrogen was not selected for the dynamic experiment because H2 adsorption on platinum exhibits slow kinetics under dynamic conditions, which would cause the pulse method to underestimate uptake relative to the volumetric reference. This distinction in adsorbate selection is itself an important data point: the choice of gas for dynamic chemisorption must be made based on the kinetic behavior of the specific metal-adsorbate system, not on general convention.
Catalyst | Pt Loading (wt%) | Method | CO Uptake (micromol/g cat.) |
|---|---|---|---|
ASTM Standard Pt/Al2O3 | 0.5 | Volumetric Chemisorption | 10.2 |
ASTM Standard Pt/Al2O3 | 0.5 | Pulse Chemisorption | 8.8 |
In-house Pt/Al2O3 | 0.3 | Volumetric Chemisorption | 10.0 |
In-house Pt/Al2O3 | 0.3 | Pulse Chemisorption | 9.1 |
The results show excellent agreement between the volumetric and pulse methods for both catalysts. The pulse method consistently yielded slightly lower uptake values than volumetric chemisorption. This systematic difference is expected and explained: the volumetric method captures weakly held or reversible CO that remains on the surface under the static equilibrium conditions of the measurement but desorbs under the inert carrier gas flow of the pulse experiment. The absolute difference is small and the trend is consistent across both catalysts.
For catalyst-adsorbate systems with fast adsorption kinetics, pulse chemisorption is a reliable and consistent method for measuring active metal surface area. The approximately 12 to 14 percent lower uptake from pulse versus volumetric methods reflects the reversible CO fraction that the volumetric technique captures. When this systematic offset is understood and accounted for in protocol design, pulse chemisorption produces data that supports the same material comparisons and ranking decisions as volumetric methods, in less than 30 minutes versus six hours or more.
Cobalt catalysts present the combination of kinetic challenges that make pulse chemisorption difficult to apply reliably. Hydrogen adsorption on supported cobalt catalysts proceeds slowly at room temperature, requiring elevation to approximately 100 degrees C for practical measurement. Carbon monoxide, the alternative adsorbate, also chemisorbs slowly on cobalt at room temperature and introduces an additional complication at elevated temperatures: CO can disproportionate via the Boudouard reaction (2CO to C + CO2), depositing carbon on the catalyst surface and distorting the measurement.
This combination makes cobalt a representative example of catalyst systems where TPD is the more appropriate dynamic technique. By pre-adsorbing hydrogen at a defined temperature and then monitoring desorption during a controlled temperature ramp, TPD avoids the kinetic limitations that would affect a pulse experiment at ambient temperature while still providing quantitative uptake data and additional qualitative information about site energy distribution.
Two cobalt catalysts supported on alumina were evaluated:
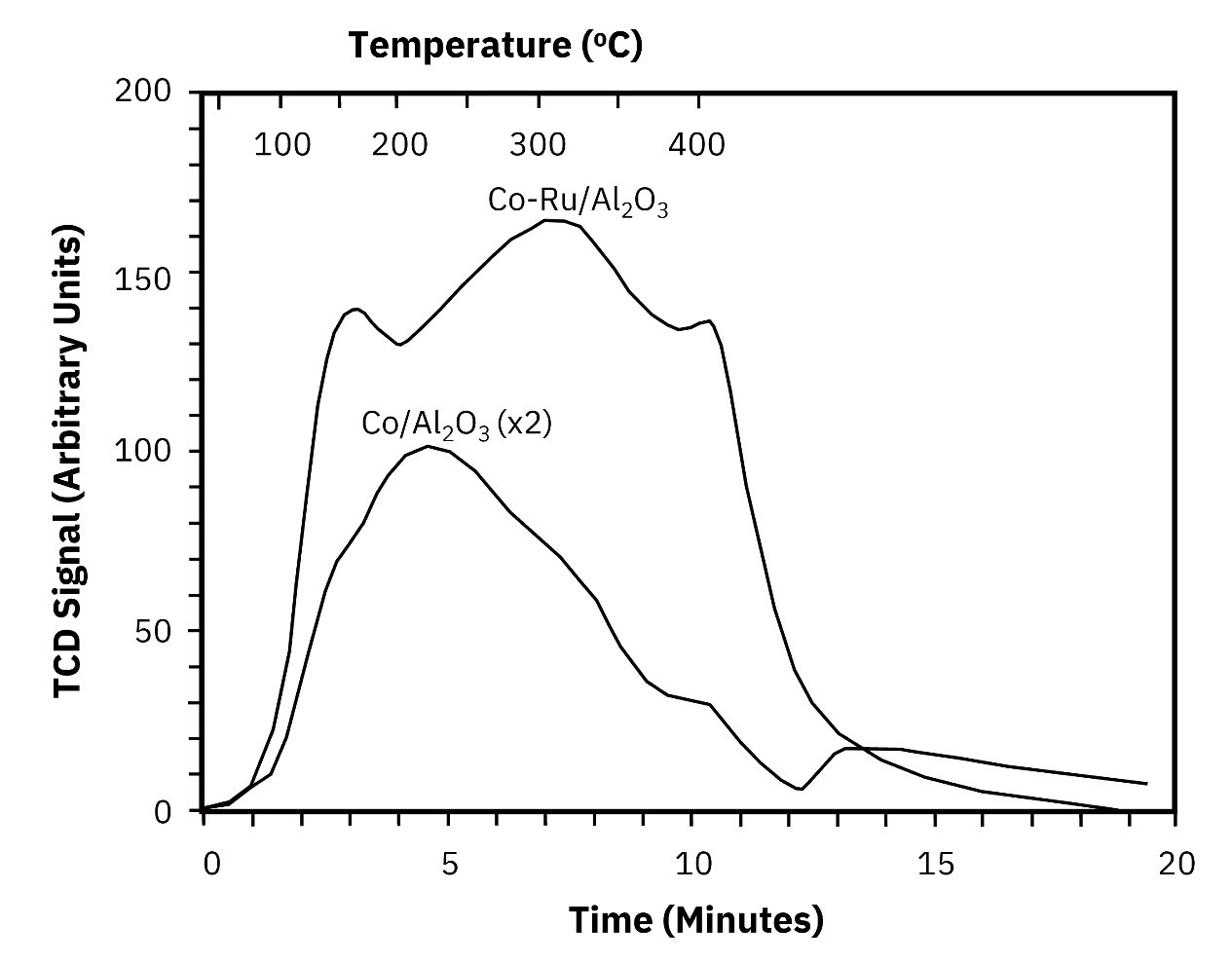
H2 was chemisorbed onto both samples at 50 degrees C for 30 minutes, followed by TPD performed up to 400 degrees C. Static H2 uptake was measured independently by a five-point volumetric method for direct comparison.
Catalyst | Promoter | Method | H2 Uptake (micromol/g cat.) |
|---|---|---|---|
20 wt% Co/Al2O3 | None | Volumetric Chemisorption | 41 |
20 wt% Co/Al2O3 | None | TPD | 42 |
0.5 wt% Ru/20 wt% Co/Al2O3 | Ru | Volumetric Chemisorption | 164 |
0.5 wt% Ru/20 wt% Co/Al2O3 | Ru | TPD | 188 |
Agreement between volumetric and TPD methods is excellent for the unpromoted Co/Al2O3 catalyst: 41 micromol/g by volumetric versus 42 micromol/g by TPD. For the Ru-promoted catalyst, the values are 164 micromol/g by volumetric and 188 micromol/g by TPD. The larger difference in the Ru-promoted catalyst is within the expected range given the different measurement conditions and the broader desorption profile shown in the TPD trace for the promoted system.
Both catalysts showed broad H2 desorption profiles in the TPD experiment, consistent with a distribution of cobalt site energies across the alumina support. The Ru-promoted catalyst produced a larger desorption signal, as expected from a catalyst with higher dispersion due to ruthenium’s role in facilitating cobalt reduction. The TPD profile shape itself, which cannot be extracted from a single-point volumetric measurement, provides this additional qualitative confirmation of the dispersion difference between the two formulations.
The key experimental parameters confirmed as essential for reliable TPD results on cobalt were an adsorption temperature of 50 degrees C and an adsorption time of 30 minutes. Deviating from these conditions would affect the completeness of surface saturation and the consistency of results across runs.
The most immediate operational benefit of dynamic chemisorption is throughput. A pulse chemisorption experiment completes in under 30 minutes after pretreatment, compared to six hours or more for a five-point volumetric measurement. For laboratories running routine catalyst QC or screening multiple synthesis conditions in a development program, this difference is substantial. A laboratory that can characterize two samples per day by volumetric methods can characterize twelve or more by pulse chemisorption on the same instrument, without needing additional equipment.
The data presented here illustrates the importance of selecting the dynamic method based on the specific catalyst-adsorbate kinetics rather than applying a single protocol universally. For Pt with CO, pulse chemisorption is fast, reliable, and gives results within 14 percent of volumetric values with a known and interpretable systematic offset. For Co with H2, TPD is the correct dynamic approach because pulse kinetics at ambient temperature are too slow. Applying pulse chemisorption to cobalt with H2 at room temperature would produce meaninglessly low uptake values that do not reflect true metal surface area.
When working with very low metal loadings, the sensitivity of pulse chemisorption can be increased without changing the instrument by using smaller pulse loops or by diluting the adsorbate in an inert carrier. Replacing pure CO with a 10 percent CO in helium mixture, for example, increases the sensitivity of TCD detection and significantly improves reproducibility. This simple protocol adjustment extends the practical applicability of pulse chemisorption to catalyst systems that would otherwise require the longer volumetric measurement to achieve adequate signal quality.
Unlike pulse chemisorption, which returns only a total uptake value, TPD provides the complete temperature profile of desorption. This profile reflects the distribution of adsorption site energies across the catalyst surface. For the cobalt catalysts in this study, the broad desorption profiles and the difference in profile shape between the promoted and unpromoted formulations provide qualitative confirmation of dispersion differences that the volumetric uptake numbers alone cannot convey. For catalyst development programs focused on understanding surface heterogeneity, this additional information is directly relevant to formulation decisions.
Reliable chemisorption data across volumetric, pulse, and TPD methods requires an instrument that handles gas switching, temperature programming, and TCD detection with the precision needed for low metal loading systems where uptake values are small and differences between catalyst formulations may be subtle.
The AMI 300 Chemisorption Analyzer is AMI’s flagship model providing fully automated static and dynamic chemisorption in a single configurable instrument. Precision gas control with independent mass flow controllers supports accurate adsorbate blending for diluted pulse experiments. A double thermocouple design ensures accurate sample temperature measurement during both adsorption and TPD experiments. Sample holders accommodate powders, pellets, extrudates, and honeycomb cores, covering the full range of supported catalyst forms.
The AMI 400 is the latest generation chemisorption analyzer, offering an automatic intelligent gas interface that selects the appropriate gas for each protocol step without manual intervention. A triple thermocouple design provides enhanced temperature control and safety. Both instruments support the complete chemisorption workflow: pretreatment, static or dynamic adsorption, and TPD, with optional in situ spectrometry integration for laboratories requiring simultaneous spectroscopic characterization.
For catalyst characterization programs that need to run both dynamic chemisorption and volumetric measurements on the same platform, the AMI 300 and AMI 400 provide that capability without requiring separate dedicated instruments for each technique.
Both dynamic chemisorption methods (pulse chemisorption and TPD) and volumetric chemisorption produce consistent and reliable results when applied to appropriate catalyst-adsorbate systems under well-controlled conditions. The CO uptake data for Pt/Al2O3 shows excellent agreement between pulse and volumetric methods, with pulse values 12 to 14 percent lower due to the reversible CO fraction that the volumetric technique captures. The H2 uptake data for Co/Al2O3 shows close agreement between TPD and volumetric methods, with TPD providing the additional benefit of site energy distribution information from the desorption profile.
The decision between dynamic and volumetric chemisorption is not about which method is more accurate in an absolute sense. It is about matching the method to the kinetic behavior of the catalyst-adsorbate system, the information required, and the throughput constraints of the laboratory. For fast-kinetics systems like CO on Pt, pulse chemisorption delivers reliable data in under 30 minutes. For slow-kinetics systems like H2 on Co, TPD provides the appropriate solution. Volumetric chemisorption remains the reference technique when reversible uptake must be captured or when a new catalyst-adsorbate system is being characterized for the first time.
Use dynamic chemisorption, either pulse or TPD, when adsorption kinetics are fast enough to achieve equilibration during the experiment and when throughput is a priority. Pulse chemisorption is the right choice for systems with rapid adsorbate kinetics, such as CO on platinum group metals at ambient temperature. TPD is the appropriate dynamic method when kinetics are slow at ambient temperature, such as H2 on cobalt, or when the adsorbate creates side reactions at the elevated temperatures needed to accelerate kinetics. Volumetric chemisorption remains the reference when weakly held or reversible adsorbate species must be quantified or when a new system is being characterized without prior kinetic knowledge.
The volumetric method measures adsorption at equilibrium under static conditions, capturing both strongly and weakly bound adsorbate species. Pulse chemisorption operates under inert carrier gas flow, which sweeps weakly held species off the surface as they desorb slowly. These desorbing molecules add to the baseline signal rather than appearing as discrete uptake events and are not counted in the pulse-by-pulse uptake sum. The result is a systematic underestimate of total uptake relative to the volumetric method, reflecting the reversible fraction of adsorbed gas. For CO on Pt/Al2O3, this difference was approximately 12 to 14 percent across the two catalysts in this study.
Pulse chemisorption returns a single total uptake value representing the quantity of gas adsorbed to saturation. TPD provides the full desorption temperature profile, which reflects the distribution of adsorption site energies across the catalyst surface. Higher desorption temperatures indicate more strongly bound species and stronger metal-support or metal-adsorbate interactions. Broader profiles indicate a wider distribution of site types. In this study, the broad H2 TPD profiles from Co/Al2O3 and the larger desorption signal from the Ru-promoted catalyst provide qualitative confirmation of dispersion differences between formulations that uptake numbers alone cannot convey. For more on this technique see our guide on temperature-programmed desorption for catalyst surfaces.
Because pulse chemisorption operates under flowing inert gas, which can:
This leads to a systematic but predictable underestimation compared to volumetric chemisorption.
Use dynamic chemisorption when:
Volumetric chemisorption is better when studying new systems or weak/reversible adsorption.
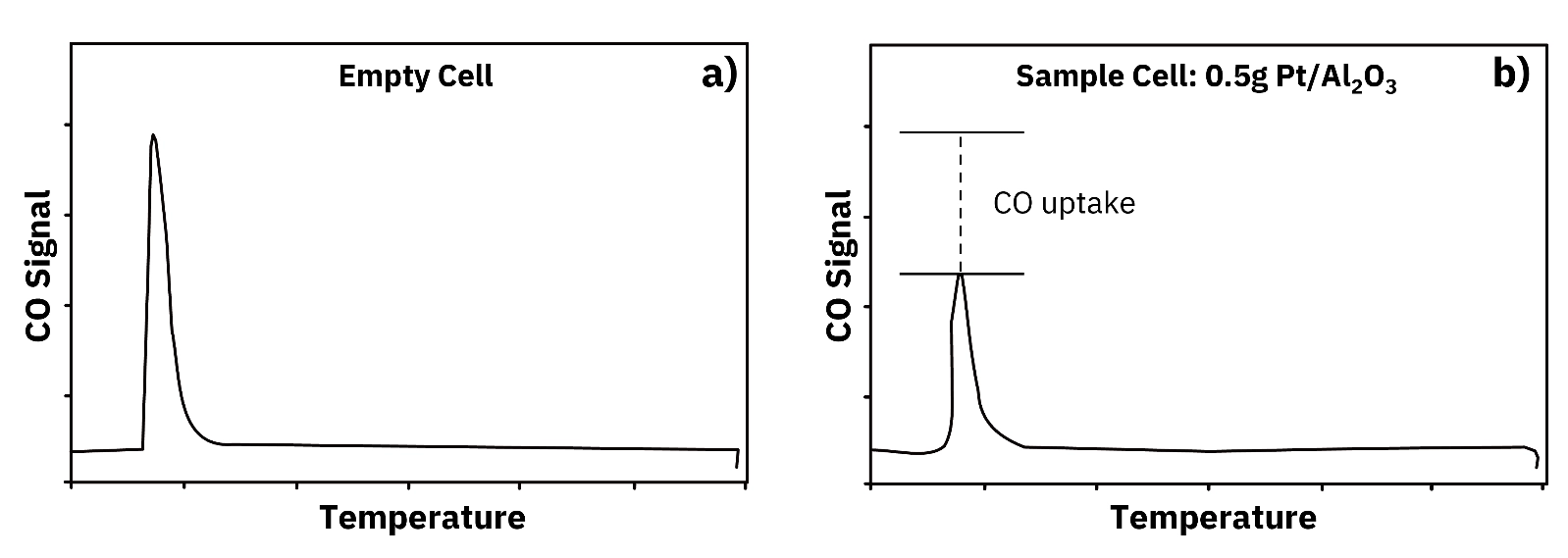
Learn how pulse chemisorption measures metal dispersion and surface area in supported catalysts, with advantages, limitations, and real published results.
Learn how N2 physisorption, TPR, and TPO deliver complete heterogeneous catalyst characterization data across zeolite, alumina, and metal oxide systems.

Learn how specific surface area and pore structure control supported ionic liquid performance in adsorption, catalysis, and heavy metal removal applications.
See how the Matrix 1000 BET analysis instrument delivers 4 alumina samples in 28 minutes with RSD below 0.3% across all ports, at production scale.