Compare dynamic chemisorption and volumetric methods for Pt and Co catalysts. See CO and H2 uptake data proving both yield consistent, reliable results.
Learn how N2 physisorption, TPR, and TPO deliver complete heterogeneous catalyst characterization data across zeolite, alumina, and metal oxide systems.
A catalyst that performs well in laboratory screening may still fail during scale-up. The pore dimensions that govern diffusion, the reduction temperatures that define activation conditions, the coke species that accumulate during operation and determine regeneration requirements: none of these are visible in reaction yield data alone. Identifying them requires characterization measurements that reach inside the material under controlled conditions. Heterogeneous catalytic processes involve cyclic repetition of elementary steps: diffusion to the surface, chemical adsorption at active sites, surface reaction, desorption, and reverse diffusion. The most critical of these are adsorption and surface reaction, and both depend on structural and chemical properties that must be measured independently to be understood. This article presents a three-technique catalyst characterization framework covering nitrogen physisorption, temperature-programmed reduction (TPR), and temperature-programmed oxidation (TPO) across five representative catalyst systems. The data demonstrates how each technique contributes information that the others cannot provide, and why integrating all three is necessary for reliable performance evaluation of heterogeneous catalysts.
Catalyst characterization is the systematic measurement of a catalyst's structural, adsorption, and reactive properties to understand how it functions at the molecular level. For heterogeneous catalysts, this means quantifying pore architecture and surface area, identifying the nature and reducibility of active sites, and evaluating deactivation behavior through coke analysis. A complete characterization program addresses three distinct dimensions:
Pore structure: Surface area, pore volume, and pore size distribution determine how reactant molecules access active sites and how products diffuse out. These are measured by nitrogen physisorption using BET and BJH or HK analysis.Active site chemistry: The reducibility of metal oxide species and their interaction with the support determine catalyst activation conditions and intrinsic reactivity. Temperature-programmed reduction (TPR) quantifies these properties.Deactivation and regeneration: Coke deposits form during catalytic reactions and block active sites. Temperature-programmed oxidation (TPO) identifies the nature of these deposits and defines the temperature conditions required to remove them.No single measurement covers all three dimensions. Treating any one of them as optional leaves critical questions unanswered before scale-up or process optimization decisions are made.The challenge in heterogeneous catalyst development is not a lack of characterization techniques. It is the tendency to apply only one or two of them and treat the resulting data as sufficient for formulation decisions. BET surface area is the most commonly measured catalyst property. It is informative, but it tells you nothing about where active metal species are located within the pore network, how strongly they interact with the support, or at what temperature they become catalytically active. Two catalysts with identical surface areas can show completely different TPR profiles, indicating fundamentally different metal-support interactions and activation requirements. Similarly, activity data from reaction testing does not identify deactivation mechanisms. When a catalyst loses performance over time, the cause may be sintering that reduces active metal dispersion, pore blockage from coke accumulation, or structural collapse under thermal cycling. Each cause requires a different corrective response. Without characterization data that separates these contributions, the decision about whether to regenerate, reformulate, or adjust operating conditions is made without sufficient information. The cost of this gap is real. In industrial settings, it means extended development cycles when catalysts fail at pilot scale for reasons that were not identified at the bench. In research programs, it limits the mechanistic insight that can be drawn from experimental data and reduces the quality of structure-property relationships that guide future synthesis decisions.
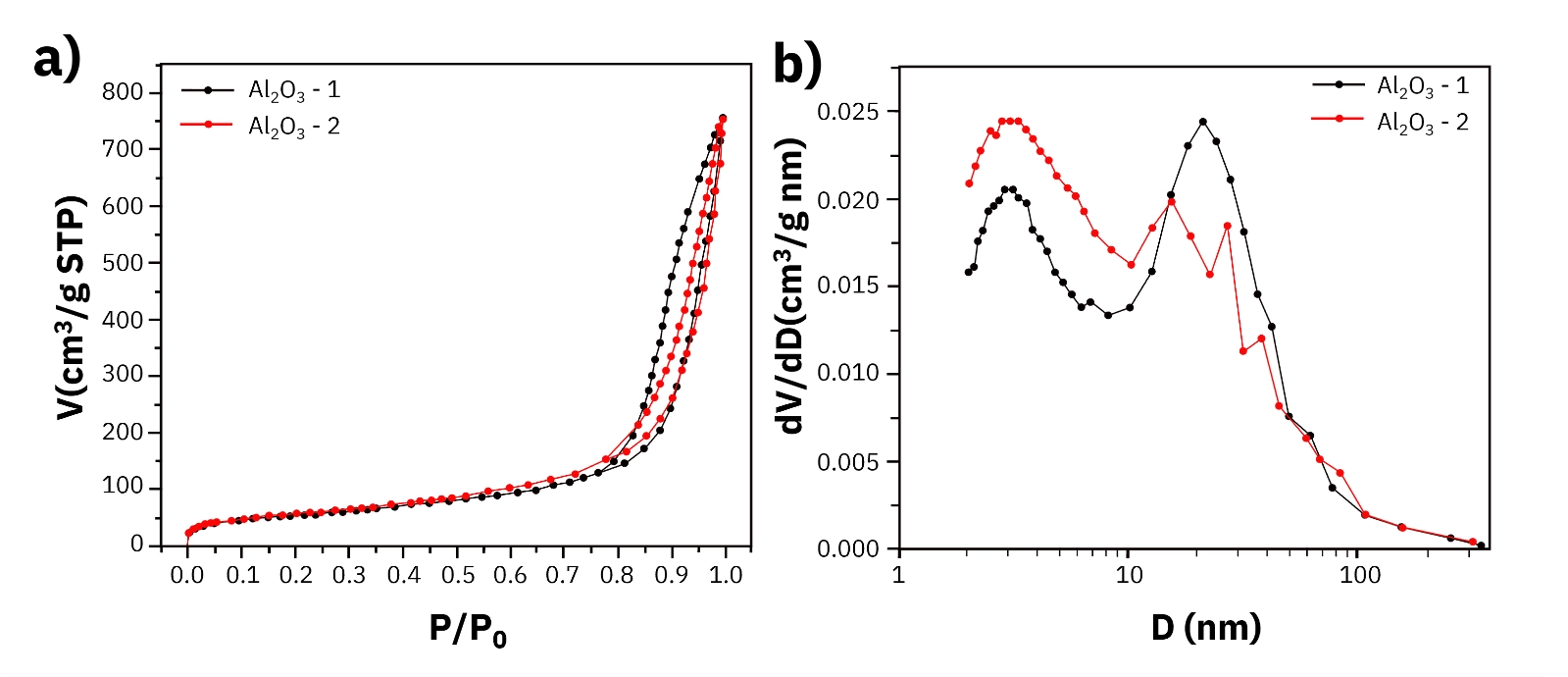
Pore Structure: Nitrogen Physisorption Across Three Catalyst Systems Nitrogen adsorption and desorption isotherms at 77 K, combined with BET, BJH, and HK analysis, provide the pore architecture data needed to understand diffusion and mass transfer in heterogeneous catalysts. Zeolite catalyst. Zeolites are microporous crystalline materials with uniform pore structures and high surface areas. The nitrogen isotherm shows a sharp uptake increase at low relative pressure, characteristic of micropore filling. HK pore size distribution analysis identifies the most probable pore diameter at 0.57 nm. BET surface area is 675 m2/g. This combination of sub-nanometer pore diameter and high surface area supports molecular sieving selectivity and high active site density per gram of material. Activated alumina. Two activated alumina samples with BET surface areas of 192.32 m2/g and 210.81 m2/g show mesopore-dominated isotherms. BJH analysis identifies pore size peaks at 3 nm and 21 nm, indicating a bimodal pore size distribution. The 3 nm population provides high surface area for active phase dispersion, while the 21 nm transport pores support diffusion of reactant and product molecules through the catalyst particle. Nickel-loaded cerium dioxide (Ni/CeO2). CeO2 is used in redox-active catalyst systems for its oxygen storage capacity and reducibility. Increasing Ni loading systematically reduces BET surface area from 15.25 m2/g at the lowest loading to 7.59 m2/g at the highest, with corresponding reductions in pore volume. This trend reflects Ni species occupying and partially blocking surface sites on the CeO2 support. Physisorption data quantifies this trade-off and provides the structural basis for interpreting the TPR results that follow. Active Site Reducibility: Temperature-Programmed Reduction TPR measures how metal oxide species in the catalyst respond to hydrogen at increasing temperature. The reduction peak temperatures indicate the strength of metal-support interaction, and the hydrogen consumption quantifies the quantity of reducible species. Alumina-supported catalyst. H2 TPR shows a single well-defined reduction peak at 234 degrees C with a hydrogen consumption of 9680 micromol/g. The single narrow peak indicates a uniform metal oxide species with a homogeneous interaction with the alumina support. The reduction temperature and hydrogen consumption together define both the activation conditions and the quantity of reducible active phase in this catalyst. Mn2O3-based catalyst. Three successive TPR peaks at 325, 440, and 597 degrees C correspond to the sequential reduction transformations: Mn2O3 to Mn3O4, Mn3O4 to MnO, and MnO to metallic Mn. Each step represents a distinct oxidation state that may be relevant to catalytic activity under different operating conditions. The well-separated peak temperatures confirm that each transformation can be accessed selectively by controlling reduction temperature, providing precise control over the active phase generated. Ni/CeO2 with increasing Ni loading. TPR profiles for 1Ni/CeO2, 2Ni/CeO2, and 3Ni/CeO2 show a primary reduction peak at 323 degrees C in all three samples, with additional peaks at 461, 464, 501, and 517 degrees C that shift and change intensity with Ni loading. Reduction temperatures increase and hydrogen consumption decreases as Ni loading rises, consistent with stronger metal-support interaction at lower loadings and a changing proportion of Ni species interacting directly with the CeO2 surface. This behavior directly connects to the surface area reduction observed in physisorption data: as Ni loading increases beyond the monolayer capacity of the CeO2 surface, metal-support interaction character changes. Coke Characterization and Regeneration: Temperature-Programmed Oxidation TPO measures the combustion temperature of carbonaceous deposits formed on the catalyst during reaction. The position and relative intensity of oxidation peaks identify the types of coke present, which determines the regeneration temperature required to restore catalyst activity. Cr2O3 catalyst after reaction. TPO data shows three oxidation peaks at 500, 578, and 631 degrees C. The dominant feature is the high-temperature peak at 631 degrees C, indicating that graphitic or deeply condensed coke species constitute the major fraction of the deposit. Lower-temperature peaks at 500 and 578 degrees C represent more reactive, lightly condensed carbonaceous species that combust more readily. The dominance of the high-temperature peak has direct implications for regenerator design: regeneration at temperatures below 631 degrees C would leave the majority of the coke deposit on the catalyst, failing to restore activity.
Shorter Development Cycles The Ni/CeO2 data illustrates the efficiency of multi-technique characterization directly. Physisorption shows that surface area decreases systematically with Ni loading. TPR shows that reduction temperature and hydrogen consumption change in parallel. Together, these two measurements define the loading-performance trade-off across the entire loading range without requiring a full reaction testing campaign at each condition. Development decisions that would otherwise require weeks of testing can be made with days of characterization data. Defined Activation and Regeneration Protocols TPR data for the alumina-supported catalyst specifies a precise activation temperature of 234 degrees C, removing ambiguity from the catalyst pretreatment protocol. TPO data for the Cr2O3 system defines a minimum regeneration temperature of 631 degrees C, ensuring that the dominant coke species is addressed in each regeneration cycle. Both specifications are derived from characterization data rather than empirical trial-and-error under process conditions. Reduced Risk at Scale-Up Catalysts that are fully characterized across pore structure, active site reducibility, and deactivation behavior before pilot-scale testing arrive with a defined operating envelope. Surface area and pore volume data quantify structural margin available for metal loading. TPR profiles confirm that activation conditions translate consistently from bench to pilot scale. TPO data validates that bench-scale regeneration protocols are effective at the temperatures achievable in pilot equipment. Reproducibility for Quality Control For catalyst producers, batch-to-batch consistency in BET surface area, pore volume, and TPR profile constitutes a quantitative quality specification. Characterization platforms that deliver reproducible data across these three measurement types support incoming material qualification and production release decisions with data rather than visual inspection or single-point activity tests.
Generating physisorption, TPR, and TPO data with the precision required for catalyst development and quality control requires instruments designed for the specific demands of each measurement type, with the gas handling, thermal control, and detection capability to produce reproducible results across diverse catalyst systems.
The AMI 300 chemisorption analyzer provides the automated gas switching, programmable temperature ramping, and TCD-based detection needed for TPR and TPO across the full range of catalyst systems shown here. Its variants include the AMI 300HP for high-pressure applications, the AMI 300IR with integrated infrared detection, and the AMI 300-SSITKA for steady-state isotopic transient kinetic analysis. For physisorption, the AMI Micro 300 delivers BET surface area, total pore volume, and pore size distribution using N2 at 77 K across the full relative pressure range required for zeolite micropore analysis, alumina mesopore characterization, and low-surface-area systems like CeO2.
Both platforms are designed for integration into a unified catalyst characterization workflow. Automated dose and evacuation controls on the Micro 300 ensure consistent physisorption results. The AMI 300’s programmable ramp rates and gas switching produce TPR and TPO profiles that are reproducible across multiple runs and operators, making the data suitable for both research reporting and quality control documentation.
For researchers and engineers working across zeolite catalysis, supported metal systems, transition metal oxides, and industrial catalyst development, this combination of physisorption and chemisorption capability covers the full characterization scope presented in this article.(1) Lv, W.; Zhang, R.; Sun, F.; Guo, Y.; Jiao, W.; Wang, H.; Hou, X.; Qin, R.; Pan, W.; Lv, B. Application of physical adsorption and chemisorption techniques in heterogeneous catalytic research: A review. Kinet. Catal. 2025, 66, 129-154. (2) Vogt, C. and Weckhuysen, B. M. The concept of active site in heterogeneous catalysis. Nat. Rev. Chem. 2022, 6, 89-111.
Catalyst characterization is the measurement of a catalyst's structural and chemical properties to understand its performance in a catalytic process. The most important techniques for heterogeneous catalysts are nitrogen physisorption (for BET surface area and pore size distribution), temperature-programmed reduction (for active site reducibility), and temperature-programmed oxidation (for coke characterization and regeneration). Each addresses a distinct dimension of catalyst behavior that the others cannot resolve. For a broader overview, see our chemisorption and catalysis reference.
BET surface area tells you how much total surface the catalyst exposes to nitrogen, but not the chemical nature or reducibility of active species on that surface. TPR identifies the temperature at which specific metal oxide species are reduced by hydrogen, the strength of metal-support interaction, and the quantity of reducible species present. For the alumina-supported catalyst in this study, TPR reveals a precise reduction peak at 234 degrees C with a hydrogen consumption of 9680 micromol/g, information that BET measurement cannot provide. See our full guide on temperature-programmed reduction parameters and profiles.
As Ni loading increases on a CeO2 support, Ni species occupy adsorption sites on the support surface, reducing the surface area accessible to nitrogen during BET measurement. At higher loadings, Ni aggregation can also partially fill pore channels, reducing total pore volume. The physisorption data presented here shows this trend quantitatively: BET surface area decreases from 15.25 m2/g at the lowest loading to 7.59 m2/g at the highest. TPR data for the same samples shows corresponding changes in reduction temperature, indicating that the structural and chemical properties of the catalyst change together as a function of Ni loading.TPO measures the combustion temperature of carbonaceous deposits by ramping temperature in the presence of oxygen. Each peak in the profile corresponds to a coke species with a specific combustion temperature. For the Cr2O3 catalyst shown, the dominant peak at 631 degrees C indicates that graphitic or deeply condensed coke constitutes the majority of the deposit. Regeneration must reach or exceed this temperature to remove most of the coke. Lower-temperature peaks at 500 and 578 degrees C represent more reactive deposits that combust earlier. See our article on advanced TPO of coked catalysts.
Multi-technique characterization is most important when characterization data will be used to make formulation decisions before reaction testing, when performance differences between catalyst variants need to be explained rather than just observed, when scale-up from bench to pilot requires defining activation and regeneration protocols with confidence, and when batch-to-batch quality control requires quantitative specifications beyond simple activity testing. For any of these use cases, the combination of physisorption, TPR, and TPO provides the structural and chemical data foundation that single-technique characterization cannot.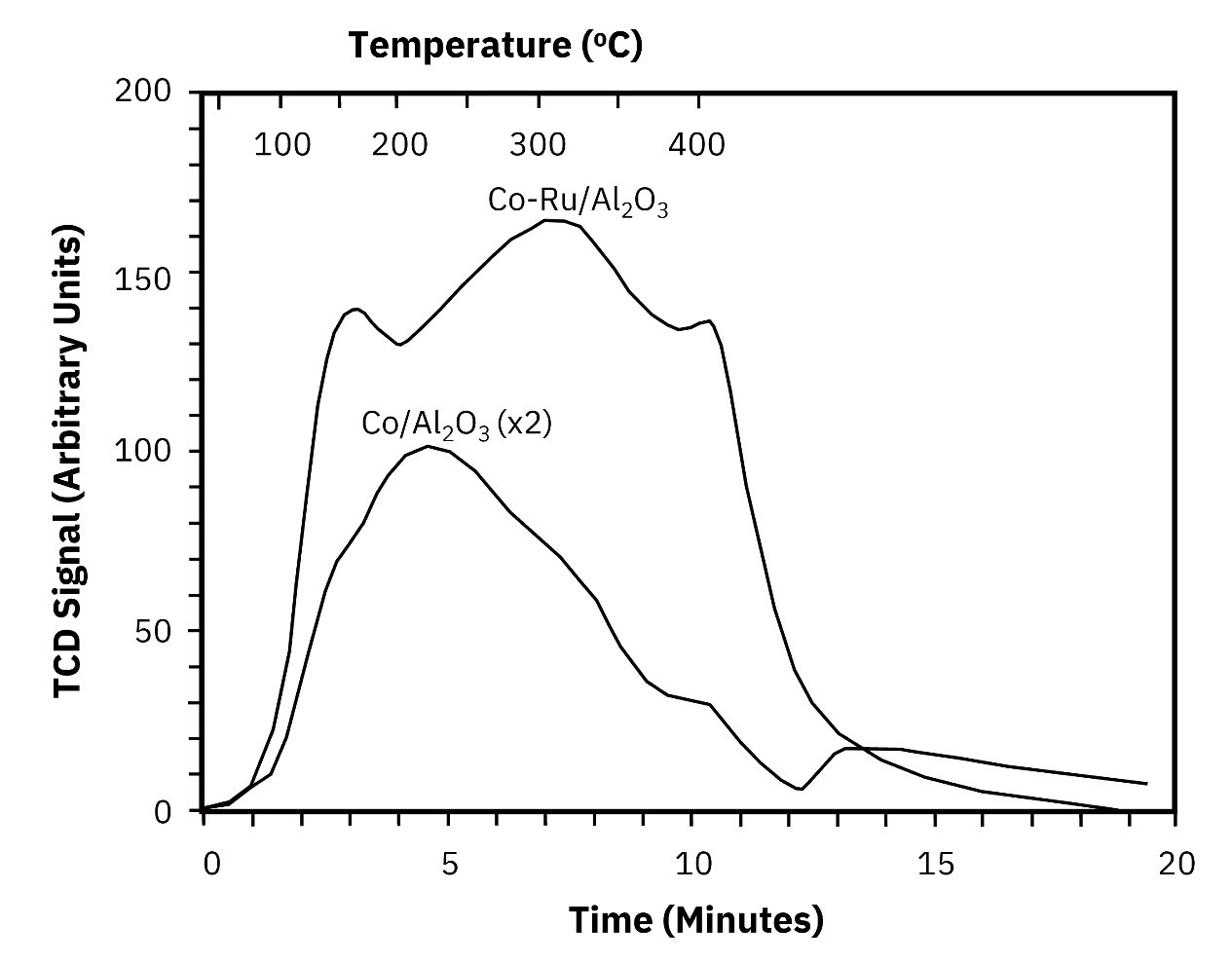
Compare dynamic chemisorption and volumetric methods for Pt and Co catalysts. See CO and H2 uptake data proving both yield consistent, reliable results.
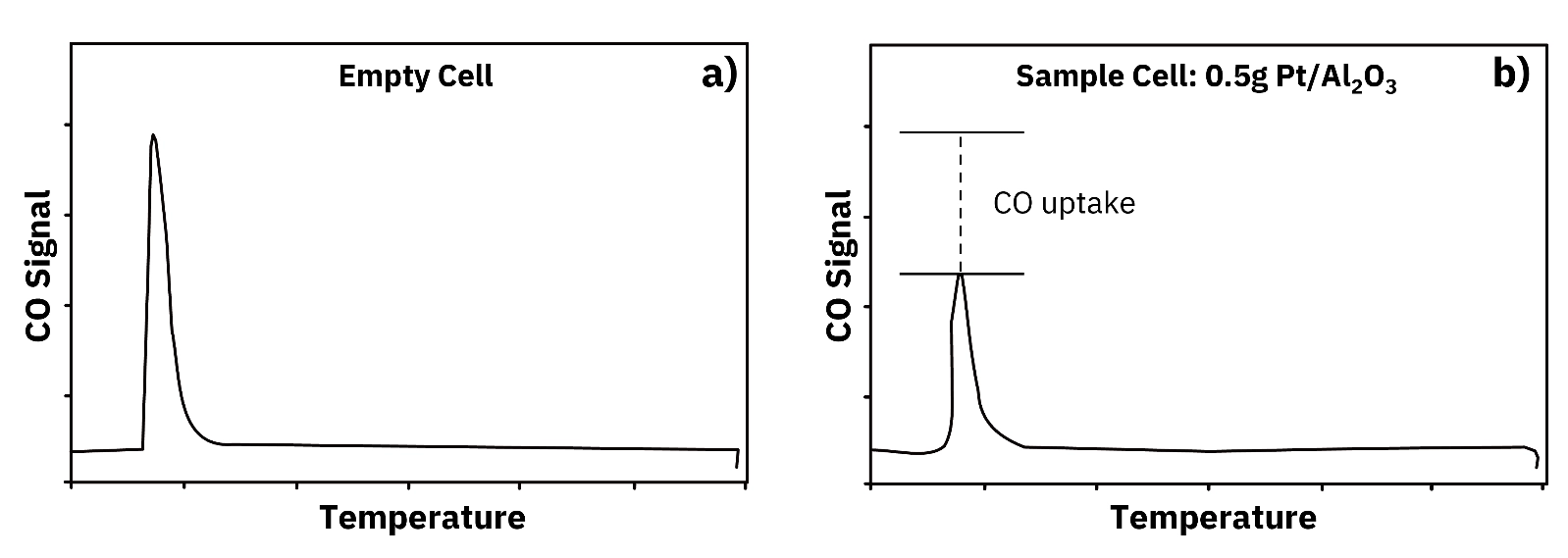
Learn how pulse chemisorption measures metal dispersion and surface area in supported catalysts, with advantages, limitations, and real published results.

Learn how specific surface area and pore structure control supported ionic liquid performance in adsorption, catalysis, and heavy metal removal applications.
See how the Matrix 1000 BET analysis instrument delivers 4 alumina samples in 28 minutes with RSD below 0.3% across all ports, at production scale.