Compare dynamic chemisorption and volumetric methods for Pt and Co catalysts. See CO and H2 uptake data proving both yield consistent, reliable results.
Metal particle dispersion is one of the most consequential structural properties of a supported metal catalyst. A highly dispersed metal phase distributes active sites across the maximum possible surface area, maximizing reaction efficiency per gram of precious or critical metal. As particles sinter and agglomerate during operation, dispersion falls, active site density drops, and conversion rate declines. The question is not whether dispersion matters. It is whether the laboratory has a reliable, fast method to measure it before and after synthesis, activation, and aging.
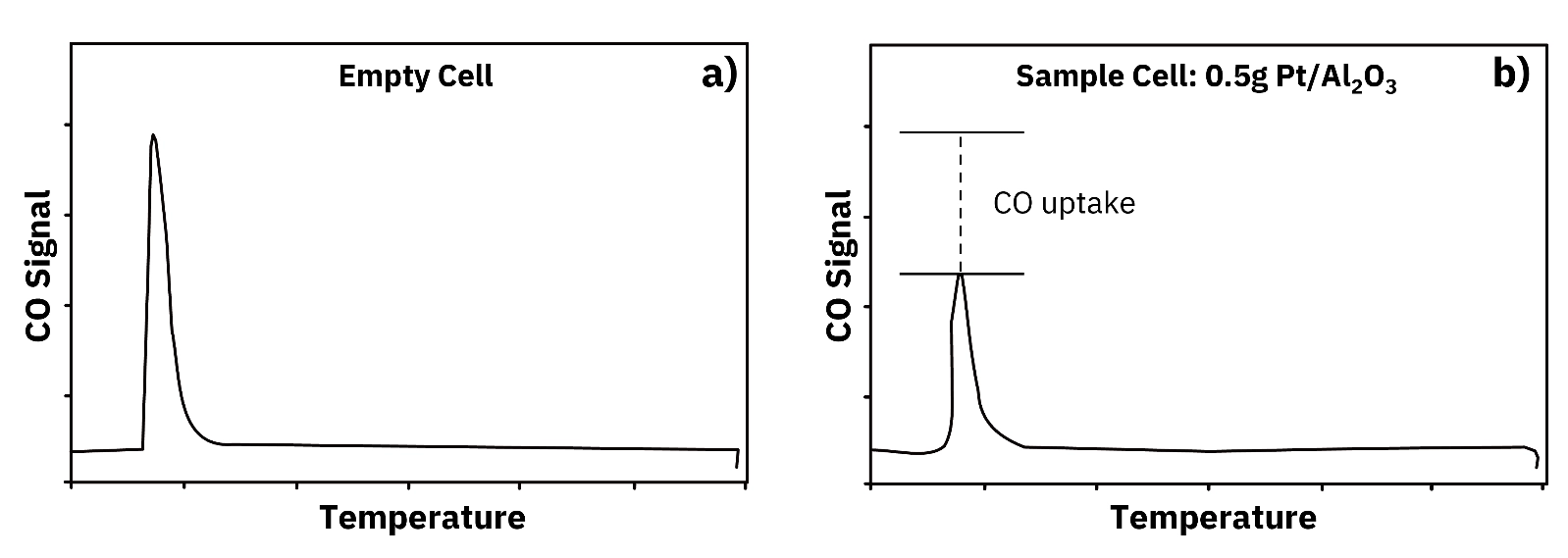
Pulse chemisorption is a flow-based technique for measuring the adsorption of a reactive gas on the surface of a reduced metal catalyst. It is used to determine two related quantities: metal particle dispersion, expressed as the fraction of total metal atoms that are exposed at the particle surface and therefore accessible for catalysis, and specific metal surface area, expressed in square meters per gram of catalyst.
These measurements require a gas that adsorbs selectively and stoichiometrically on the metal of interest without adsorbing on the oxide support. Carbon monoxide (CO) and hydrogen (H2) are the most common choices. CO equilibrates quickly and binds strongly to most metals, making it well-suited for measuring active site density on platinum, palladium, rhodium, and other platinum group metals. H2 is effective across a broad range of metals and is non-toxic, which simplifies laboratory handling. N2O is also used for specific systems, particularly for measuring Cu0 surface area where its selective reaction with copper avoids interference from oxide support surfaces.
The result of a pulse chemisorption experiment is a quantitative measurement of accessible metal surface area based on the actual chemisorption behavior of the catalyst under controlled conditions, not an estimate derived from particle size assumptions.
Determining metal dispersion and surface area in supported catalysts is not straightforward. The metal phase is present at low concentrations on a high-surface-area support, and the measurement must distinguish adsorption on the metal from any weak adsorption on the support material. Several specific challenges make this measurement sensitive to experimental conditions:
Sample pretreatment directly determines the oxidation state of the metal surface at the time of measurement. Metal that has not been fully reduced will show lower apparent dispersion than a fully reduced sample, not because dispersion has changed but because fewer metal sites are accessible to the probe gas
Weakly adsorbed species present a quantification problem in flow-based techniques. Gas that adsorbs weakly desorbs slowly under the inert carrier flow, adding to the baseline signal rather than appearing as a distinct uptake event. This leads to underestimation of total chemisorption uptake
Slow adsorption kinetics at the measurement temperature can produce the same underestimation effect. If the carrier flow sweeps gas past the catalyst faster than the metal surface can achieve equilibrium adsorption, the measured uptake will be lower than the true value
Different adsorbate gases give different stoichiometries on the same metal, meaning that the choice of CO versus H2 versus N2O affects the calculated dispersion value even for the same catalyst. Selecting the appropriate gas and stoichiometry requires knowledge of the metal-adsorbate system
These challenges do not make pulse chemisorption unreliable. They make it sensitive to experimental protocol. When the technique is applied with appropriate pretreatment, correct gas selection, and an instrument capable of precise pulse delivery and TCD detection, the results are quantitative and reproducible.
Pulse chemisorption measurements are carried out at atmospheric pressure in a flow system at ambient temperature. A small catalyst sample, typically 0.1 to 1.0 g, is placed in a small-diameter cell of quartz or stainless-steel construction. After appropriate pretreatment to produce a clean, reduced metal surface, the sample is subjected to flow of an inert carrier gas, typically argon.
A small, precisely defined volume of chemisorbing gas is injected as a pulse into this argon stream before it flows over the catalyst. The composition of the gas exiting the sample cell is monitored continuously by a thermal conductivity detector (TCD). The signal produced when a pulse passes through the sample cell is compared to the signal produced by an identical pulse through an empty cell. The difference between the two signals represents the quantity of gas adsorbed on the catalyst surface during that pulse.
Over a clean, fully reduced catalyst surface, the initial pulse is almost entirely adsorbed. Very little signal appears at the detector after pulse one, indicating complete uptake. The procedure is then repeated. With each subsequent pulse, the catalyst surface becomes progressively more saturated with adsorbate, and more gas passes through to the detector. The detector signal increases with each pulse until it becomes constant and equal to the blank cell signal, indicating that the surface is fully saturated. The total chemisorption uptake is the sum of all uptakes from each pulse prior to saturation.
Figure 2 in the source document illustrates this with a 50-microliter pulse of CO in helium: the pulse passing through an empty cell produces a full, symmetric signal peak, while the same pulse through a cell containing 0.5 g of Pt/Al2O3 shows a substantially reduced peak, with the difference corresponding to CO adsorbed by the platinum surface. Figure 3 shows the full pulse sequence for Pt/Al2O3, from the near-zero signal of pulse 1 through the progressive saturation of pulses 2, 3, and beyond until pulses 4 and 12 overlap, indicating complete surface saturation.
Pulse chemisorption is faster and simpler to execute than most other selective chemisorption methods. Its practical advantages over volumetric chemisorption and temperature-programmed desorption (TPD) include:
These advantages explain why pulse chemisorption is often the preferred method for determining metal surface areas in both research and production catalyst development environments.
Recognizing the limitations of pulse chemisorption is as important as understanding its advantages. Flow-based techniques including pulse chemisorption and TPD systematically give lower uptake values than volumetric chemisorption for two reasons.
First, weakly held adsorbate molecules are difficult to quantify under inert gas flow. When a weakly adsorbed species desorbs slowly and continuously under the carrier stream, it adds to the baseline signal rather than appearing as a discrete uptake event. This contribution is not captured in the pulse-by-pulse uptake calculation and leads to underestimation of total chemisorption.
Second, slow adsorption kinetics at the measurement temperature can prevent complete equilibration during the brief contact time of each pulse. Gas that does not adsorb within the residence time of the pulse is swept past the catalyst and detected as unadsorbed, again leading to underestimation.
Both effects are most significant for catalyst systems with weak metal-adsorbate interactions or at measurement temperatures where kinetics are slow. For strongly adsorbing systems such as CO on platinum group metals at ambient temperature, these effects are minimal and pulse chemisorption results agree closely with volumetric measurements. For systems where the metal-adsorbate interaction is weaker, the choice of technique and adsorbate gas requires more careful consideration.
Two recent studies demonstrate the application of pulse chemisorption using AMI Instruments systems to answer specific catalyst development questions.
Kanuri et al. (2025) used the AMI 300 Chemisorption Analyzer with H2 adsorbate to calculate the dispersion and metallic surface area of Cu0 in CuO-ZnO-CeO2 catalysts prepared by different synthesis methods. Combined with X-ray diffraction and scanning electron microscopy, H2 pulse chemisorption provided the quantitative metal dispersion data needed to determine which synthesis approach produced the highest Cu dispersion. This is a direct application of pulse chemisorption to a materials development decision: choosing between synthesis routes based on the dispersion of the active copper phase rather than on bulk compositional analysis alone.
Warmuth et al. (2026) used the AMI 300 with N2O gas to calculate the Cu0 surface area of catalysts supported on ZnO/ZrO2 and ZnO/ZrO2/SiO2. By measuring Cu0 surface area as a function of reaction time on stream, the study quantified how catalyst stability varies with support composition under operating conditions relevant to methanol synthesis from green hydrogen. This application shows pulse chemisorption in a deactivation monitoring role: tracking the loss of active metal surface over time to compare support formulations.
In both cases, pulse chemisorption provided the specific, quantitative metal surface area data that complementary structural techniques could not deliver independently.
The CuO-ZnO-CeO2 study illustrates the direct value of dispersion measurement in formulation decisions. When multiple synthesis methods are being evaluated for a new catalyst, the relevant question is which method produces the highest active metal dispersion at the intended metal loading. Pulse chemisorption answers this question in less than an hour per sample, enabling comparison across synthesis routes without committing to full reaction testing at each condition.
The methanol catalyst study demonstrates how pulse chemisorption supports stability assessment. Measuring Cu0 surface area at intervals during a reaction run quantifies the rate of active site loss, making it possible to compare support formulations on the basis of measured deactivation rate rather than observed activity decline alone. This distinction matters for support selection decisions where two formulations may show similar initial activity but different long-term stability.
For catalyst manufacturers, batch-to-batch consistency in metal dispersion is a quality specification, not just a research metric. Pulse chemisorption’s speed, fewer required parameters, and ease of standardization make it practical as a production quality control measurement. A fully reduced reference catalyst measured on a standardized protocol provides a reproducible baseline against which each new batch can be compared in less than an hour.
For catalysts based on platinum group metals, higher dispersion means more active sites per gram of metal. Characterization data that identifies formulations with superior dispersion supports the rational reduction of precious metal loading without compromising activity. This has a direct and measurable impact on catalyst cost per unit of production capacity.
Generating reliable pulse chemisorption data requires an instrument that delivers precise pulse volumes, stable carrier gas flow, accurate TCD detection, and the automated gas handling needed to execute multi-step pretreatment and measurement protocols without operator error.
The AMI 300 Chemisorption Analyzer is AMI’s flagship model for fully automated static and dynamic chemisorption. It features precision gas control with independent mass flow controllers for gas blending, a double thermocouple design for accurate temperature measurement, and sample holder options supporting powders, pellets, extrudates, and honeycomb cores. The AMI 300 has been used in multiple published studies for pulse chemisorption, including the CuO-ZnO-CeO2 and methanol catalyst work referenced in this article, confirming its performance in active research programs.
The AMI 400 is AMI’s latest generation fully automated and customizable chemisorption analyzer. It features an automatic intelligent gas interface that selects the appropriate gas for each step without manual switching, precision gas control with independent mass flow controllers, and a triple thermocouple design for enhanced temperature control and safety. The AMI 400 offers enhanced safety features and broader customizability relative to the AMI 300, making it suited for laboratories with more demanding safety requirements or complex multi-gas protocols.
Both instruments support the full range of pulse chemisorption adsorbates, including CO, H2, and N2O, across the catalyst systems most relevant to current research and industrial development.
CO and H2 are the most common choices. CO adsorbs strongly and equilibrates quickly on most metals, making it well-suited for platinum group metals. H2 is effective and non-toxic, which simplifies handling. N2O is used for selective measurement of Cu0 surface area, where it reacts with copper without adsorbing on oxide supports. The choice depends on the metal, the support chemistry, and the measurement temperature. Using an inappropriate gas or stoichiometry will produce a dispersion value that does not reflect the true accessible metal surface.
Pulse chemisorption is faster and simpler. It can be completed in less than an hour with only three parameters to set: flow rate, temperature, and pulse size. Volumetric chemisorption requires longer equilibration periods and more complex experimental protocols. However, flow-based techniques including pulse chemisorption systematically underestimate uptake relative to volumetric methods for weakly adsorbing systems, because weakly bound species desorb under the carrier flow and slow kinetics prevent full equilibration during each pulse contact time. For strongly adsorbing systems at ambient temperature, the two techniques agree closely. See our comparison of static and dynamic chemisorption methods.
Metal dispersion is the ratio of surface metal atoms, those accessible to the chemisorbing gas, to total metal atoms in the sample. It is calculated from the total chemisorption uptake across all pulses before saturation, combined with the known stoichiometry of the metal-adsorbate interaction (how many adsorbate molecules adsorb per surface metal atom) and the metal loading of the catalyst. Higher dispersion means more surface metal atoms per gram of total metal, which generally corresponds to smaller, more uniformly distributed metal particles. For more on the methodology, see our article on measuring metal dispersion by pulse chemisorption.
The AMI 300 is AMI’s model with fully automated static and dynamic chemisorption capability, precision mass flow controllers, and a double thermocouple design. It has a proven track record in published research and supports all standard chemisorption protocols. The AMI 400 is the latest generation instrument with an automatic intelligent gas interface that selects the appropriate gas at each step without manual intervention, a triple thermocouple design for enhanced temperature control and safety, and greater customizability. The AMI 400 is suited for laboratories with more complex multi-gas protocols or stricter safety requirements.
Extremely important. Pretreatment (typically reduction) ensures the metal is in its active state. Poor reduction leads to:
Compare dynamic chemisorption and volumetric methods for Pt and Co catalysts. See CO and H2 uptake data proving both yield consistent, reliable results.
Learn how N2 physisorption, TPR, and TPO deliver complete heterogeneous catalyst characterization data across zeolite, alumina, and metal oxide systems.

Learn how specific surface area and pore structure control supported ionic liquid performance in adsorption, catalysis, and heavy metal removal applications.
See how the Matrix 1000 BET analysis instrument delivers 4 alumina samples in 28 minutes with RSD below 0.3% across all ports, at production scale.