Compare dynamic chemisorption and volumetric methods for Pt and Co catalysts. See CO and H2 uptake data proving both yield consistent, reliable results.
Learn how a breakthrough curve analyzer helps evaluate CO2 capture in MEA liquid absorbents using BTsorb 100, with repeatability and regeneration insights.

Decreasing our CO2 emissions remains one of the hardest tasks in industrial de-carbonization. Among the key carbon capture methods, liquid absorption remains a pretty practical option because it combines a very effective CO2 uptake with lots of existing industry knowledge. In quite a few systems, amine-based solvents are often used - they give us just the right balance of reactivity, efficiency, and scalability. One of the most broadly used solvents hereabouts is monoethanolamine - or MEA. It really is quite popular since it reacts very fastly indeed with CO2, offers a great deal of absorption performance itself, and also happens to be relatively less expensive overall. However, at the same time, there are some give-and-take elements involved. A bit more energy is actually necessary during regeneration, and the solvent itself might degrade over time slightly if we operate under certain specific conditions. That's the reason why really trustworthy dynamic testing really counts so much more when assessing the actual capture performance in real life. For labs studying process-relevant adsorption behavior, a BTsorb 100 breakthrough analyzer can help measure breakthrough time, dynamic uptake, and mass-transfer behavior under controlled conditions. The BTsorb 100 Series is positioned by AMI as a dynamic sorption system for breakthrough testing, competitive adsorption, and mass transfer kinetics analysis, including applications in CO2 capture.
Liquid absorbents continue to matter because they can handle high CO2 loads and are already familiar in many industrial and pilot-scale setups. While other capture routes such as membranes and solid adsorbents are also important, liquid absorption remains a practical choice when researchers need a well-known chemistry platform for testing and comparison. In a typical liquid absorption process, CO2 first moves from the bulk gas phase toward the gas film, then crosses the gas-liquid interface, dissolves into the liquid, and finally diffuses into the bulk liquid where it reacts with the solvent. This sequence is important because it shows that performance is not only about equilibrium capacity. Transport behavior, diffusion, and mass transfer also shape the final result. That is exactly why dynamic tools are useful. Instead of showing only a static endpoint, breakthrough testing helps researchers see how the system behaves as gas flows through it over time. For teams working across a broader characterization workflow, AMI also offers gas adsorption instruments that support adsorption and separation studies across different materials and applications.
MEA is often seen as a standard absorbent benchmark - it offers a high degree of reactivity, quite rapid absorption kinetics, and an impressive CO2 holding capacity per unit mass. Additionally, it's widely available commercially which really helps with comparing lab findings to known performance ranges. In the original paper, the anticipated commercial capacity is actually stated as approximately 0. 5 moles of CO2 per mole of amine, giving researchers a really helpful reference point when they're looking at test results.To study CO2 absorption in MEA, the article used the BTsorb 100 breakthrough curve and mass transfer analyzer with a commercially available MEA solution. A condenser was installed at the sample cell outlet and maintained at 0 °C to limit interference from MEA vapor, while a desiccant in the cold trap helped remove residual moisture from the gas stream. Before each experiment, the system was purged with nitrogen at 185 mL/min to remove any remaining CO2 from the setup. The testing included fresh MEA, regenerated-and-reused MEA, and a second fresh MEA sample to check repeatability. Regeneration was carried out by heating the sample cell to 120 °C, and once outlet CO2 concentration dropped below 0.5%, absorption was repeated under the same conditions. This approach made it possible to compare fresh performance against post-regeneration behavior in a direct and practical way.

This kind of setup matters because breakthrough analyzers are designed to show dynamic behavior, not just a final uptake value. According to AMI’s BTsorb 100 product page, the system supports breakthrough curve analysis, competitive adsorption, and diffusion-related studies, making it suitable for labs that want more process-relevant insight into gas separation and CO2 capture performance.The article reports that the two fresh MEA tests produced nearly identical breakthrough curves. That is a good sign because it shows strong repeatability in the experimental setup. When a repeated fresh sample behaves almost the same way, researchers can feel more confident that later differences are coming from the sample condition, not from unstable test conditions. After regeneration, the breakthrough time became shorter. That points to a drop in absorption performance. The measured absorption capacities for the two fresh 10 mL MEA samples were 0.4875 mol/mol and 0.4822 mol/mol, which align closely with the commonly reported commercial value of about 0.5 mol CO2 per mol amine. After desorption and re-absorption, the capacity decreased to 0.3875 mol/mol. The article attributes this drop to likely causes such as partial thermal degradation or incomplete recovery of active absorption sites.
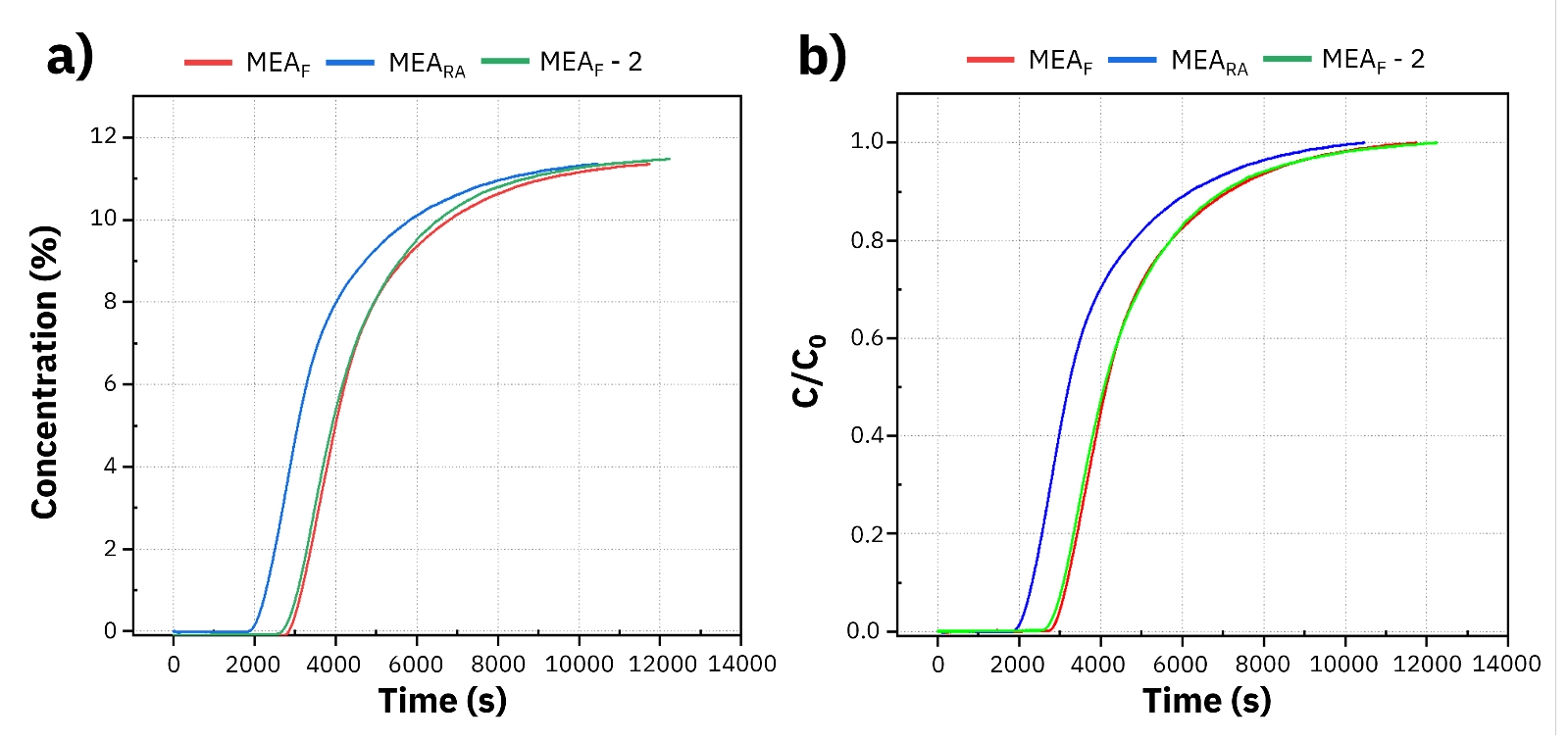
These results are useful because they do more than confirm that MEA works. They also show why regeneration performance needs to be part of the evaluation. A solvent can still look strong in a first-pass test while losing efficiency after thermal cycling. That is exactly the kind of detail dynamic breakthrough testing helps reveal. The AMI technical library version of this article presents the same main conclusion, noting the strong fresh performance and the clear decline after regeneration.A static measurement can tell you how much gas a system can hold under a defined condition, but it does not always show how the capture process unfolds in real time. A breakthrough curve analyzer fills that gap by tracking how fast the gas front moves, when saturation begins, and how the system responds under flow conditions. That makes it especially useful for process-oriented studies where timing, mass transfer, and dynamic behavior all matter. The article also highlights another practical strength of the BTsorb 100 workflow: it supports condensers, cold traps, and in-line conditioning, which helps when testing challenging streams such as moisture-bearing or reactive gases. On the product side, AMI describes the BTsorb 100 Series as suitable for evaluating adsorption and separation in porous materials and lists CO2 capture among its application areas. For teams that want a wider picture of sorption behavior, this dynamic data can be paired with other characterization tools. The article itself points to related static and gravimetric datasets, and labs that need deeper pore or surface-related adsorption insight can complement dynamic testing with the Micro 300 micropore analyzer. For higher-pressure capacity studies, the RuboSorp MPA high-pressure sorption system can extend the analysis beyond standard breakthrough conditions.
This study shows that MEA remains a strong candidate for CO2 capture in liquid absorption systems, especially when researchers need a familiar and commercially relevant solvent for comparison. It also shows that fresh performance is only part of the story. Regeneration behavior matters, and a clear drop after re-absorption can reveal limits that would be easy to miss in a simpler test design. For labs that want to evaluate dynamic transport, repeatability, and cycle performance in a more realistic way, a breakthrough curve analyzer can provide more useful insight than a capacity number alone. That makes it a practical tool for researchers working on solvent screening, kinetic evaluation, and carbon capture process development.

(1) Osman, A. I.; Hefny, M.; Abdel Maksoud, M. I. A.; Elgarahy, A. M.; Rooney, D. W. Recent advances in carbon capture storage and utilisation technologies: A review. Env. Chem. Lett. 2021, 19, 797-849. (2) Chowdhury, F. A.; Yamada, H.; Higashii, T.; Goto, K.; Onada, M. CO2 capture by tertiary amine absorbents: A performance study. Ind. Eng. Chem. Res. 2013, 52, 8323-8331. (3) Rao, A. B. and Rubin, E. S. A technical, economic, and environmental assessment of amine-based CO2 capture technology for power plant greenhouse gas control. Environ. Sci. Technol. 2022, 36, 4467- 4475. (4) Wang, N.; Wang, D.; Krook-Riekkola, A.; Ji, X. MEA-based CO2 capture: A study focuses on MEA concentrations and process parameters. Front. Energy Res. 2023, 11, 1230743. (5) de Ávila, S. G.; Logli, M. A.; Matos, J. R. Kinetic study of the thermal decomposition of monoethanolamine (MEA), diethanolamine (DEA), triethanolamine (TEA), and methyldiethanolamine (MDEA). Int. J. Greenhouse Gas Control, 2015, 42, 666-671. (6) Ge, Y.; Zhang, Z.; Yang, L.; Liu, F.; Yang, X.; Liu, K. Surfactant-modified monoethanolamine for better foaming to enhance CO2 removal efficiency. Chem. Eng. J. 2024, 498, 155440. (7) Wang, Y.; Cheng, F.; Li, J.; Liu, Y.; Wang, H.; Li, Z.; Yang, X. Development of MEA-based and AEP based CO2 phase change absorbent. Processes, 2016, 13, 92. (8) Dugas, R. and Rochelle, G. Absorption and desorption rates of carbon dioxide with monoethanolamine and piperazine. Energy Procedia, 2009, 1, 1163-1169.
A breakthrough curve analyzer is used to study how gases move through a system over time under flow conditions. It helps researchers measure breakthrough time, dynamic uptake, and mass-transfer behavior instead of relying only on a final equilibrium value.
MEA is widely used because it reacts quickly with CO2, has strong absorption performance, and is already established in commercial and pilot-scale carbon capture systems. It is also useful as a benchmark solvent because its expected performance range is well known.
In the article’s results, regenerated MEA showed lower absorption capacity than fresh MEA. The likely reasons were partial thermal degradation during desorption or incomplete recovery of active absorption sites after regeneration.
A breakthrough curve analyzer supports CO2 capture research by providing real-time data on how carbon dioxide moves through and interacts with liquid absorbents like MEA. It helps measure breakthrough time, dynamic absorption capacity, and mass transfer behavior, giving a more accurate picture of performance under realistic flow conditions.
Regeneration performance is important because it determines whether the solvent can maintain its CO2 absorption efficiency over multiple cycles. As shown in the study, regenerated MEA often shows reduced capacity due to thermal degradation or incomplete recovery, making cycle testing essential for evaluating long-term performance.
Compare dynamic chemisorption and volumetric methods for Pt and Co catalysts. See CO and H2 uptake data proving both yield consistent, reliable results.
Learn how pulse chemisorption measures metal dispersion and surface area in supported catalysts, with advantages, limitations, and real published results.
Learn how N2 physisorption, TPR, and TPO deliver complete heterogeneous catalyst characterization data across zeolite, alumina, and metal oxide systems.

Learn how specific surface area and pore structure control supported ionic liquid performance in adsorption, catalysis, and heavy metal removal applications.