Learn how BET and t-range selection affects carbon black surface area accuracy, with NSA and STSA data across 11 standard carbon black grades.
Explore how a MOF-based direct air capture CO2 adsorbent performs across pore characterization humid breakthrough testing with AMI Instruments.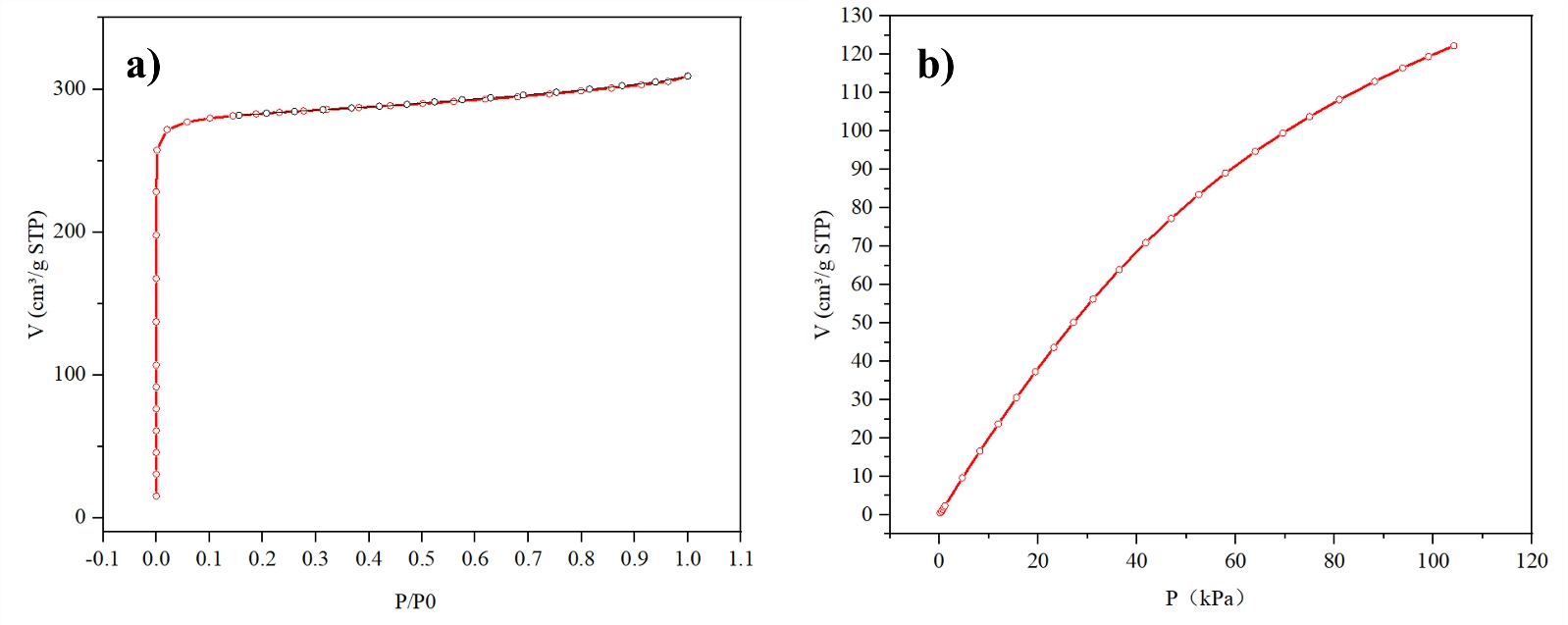
Direct air capture (DAC) is one of the few carbon removal technologies capable of addressing emissions that cannot be eliminated at the source. But capturing CO2 from ambient air is analytically and materially demanding in ways that distinguish it from conventional carbon capture from industrial flue gas. Understanding how a candidate adsorbent performs under realistic DAC conditions requires a measurement strategy that goes beyond standard surface area analysis. This article presents a multi-method characterization study of a MOF-based adsorbent evaluated for DAC relevance using nitrogen physisorption, CO2 adsorption isotherms, thermogravimetric analysis, and breakthrough curve testing under humid conditions.
A direct air carbon capture is a solid material that selectively captures carbon dioxide from ambient air, where CO2 is present at a concentration of approximately 400 parts per million and a partial pressure of approximately 40 Pa. This is fundamentally different from point-source capture, where flue gas CO2 partial pressures can reach approximately 12 kPa, providing a much stronger driving force for adsorption. Effective DAC adsorbents must meet several requirements simultaneously: high CO2 adsorption capacity, fast adsorption kinetics, strong selectivity for CO2 over nitrogen and water vapor, and stability across repeated adsorption and regeneration cycles. Solid sorbents, including metal-organic frameworks (MOFs), supported amines, and alkali or alkaline-earth materials, are preferred over liquid amine solutions for DAC because they avoid problems associated with solvent volatility, corrosion, and high energy consumption during regeneration.
Evaluating a DAC adsorbent is more demanding than routine surface area or pore size characterization. Several overlapping technical challenges define what a complete characterization program must address:CO2 partial pressure in ambient air is approximately 300 times lower than in typical industrial flue gas, meaning adsorption behavior must be evaluated at very low driving forces where standard isotherm measurements may not capture the relevant performance windowWater vapor in ambient air competes with CO2 for adsorption sites on hydrophilic surfaces, and this competitive effect can substantially reduce effective CO2 capacity under real operating conditionsPore structure governs adsorption potential energy at low pressures, making detailed micropore characterization a necessary input for understanding capacity and selectivityA material that shows strong CO2 uptake under pure-gas or dry conditions may perform very differently at 400 ppm CO2 in a humid air stream, and only dynamic breakthrough testing under those conditions can quantify the differenceMeeting all of these measurement requirements with a single instrument or technique is not possible. A comprehensive DAC adsorbent evaluation requires a coordinated set of analytical methods that together cover pore structure, equilibrium capacity, dilute-gas uptake, and dynamic performance under humidity.At the very low CO2 partial pressures encountered in DAC applications, the interaction between CO2 molecules and the adsorbent surface is the primary factor controlling uptake. In ultramicropores, pores with widths below approximately 0.7 nm, the adsorption potential energy field from opposing pore walls overlaps and creates a substantially stronger adsorption environment than in larger pores. This enhancement is particularly significant at low relative pressures, making ultramicropore volume a key structural parameter for DAC-relevant performance. BET surface area provides a measure of total accessible surface, but pore size distribution and micropore volume give more direct insight into the fraction of the pore network that is active at the pressures relevant to ambient CO2 capture. For this reason, nitrogen physisorption at 77 K is the appropriate starting point for DAC adsorbent characterization, providing the structural baseline that contextualizes all subsequent performance measurements.
This study used three AMI Instruments systems, each addressing a distinct aspect of DAC adsorbent evaluation. AMI Matrix 1000: Pore Structure and CO2 Isotherm The AMI Matrix 1000 specific surface area and pore size analyzer was used to collect nitrogen adsorption and desorption isotherms at 77 K and CO2 adsorption isotherms at 25 degrees C (298 K). The nitrogen data provided BET surface area, micropore volume, and pore size distribution. The CO2 isotherm data established the equilibrium adsorption capacity at pressures up to 100 kPa, providing the upper bound for adsorption performance under idealized conditions. AMI TGA 1000: Uptake Under Pure and Dilute CO2 The AMI TGA 1000 thermogravimetric analyzer was used to measure CO2 uptake by tracking sample mass change during controlled gas exposure. Two conditions were evaluated: pure CO2 at 50 mL/min with pretreatment at 80 degrees C followed by adsorption at 25 degrees C, and a 400 ppm CO2 in N2 mixture at 50 mL/min with the same pretreatment and adsorption temperatures. The dilute-gas condition mirrors the CO2 concentration in ambient air and provides a direct measurement of DAC-relevant uptake under dry conditions. AMI BTsorb 100: Dynamic Breakthrough Testing Under Humidity The AMI BTsorb 100 penetration curve and mass-transfer analyzer was used for breakthrough testing at conditions designed to simulate ambient DAC operation: 400 ppm CO2, 40% relative humidity, 10 mL/min flow rate, 298 K, and 1 bar. Breakthrough profiles were used to calculate dynamic adsorption capacity, which reflects how the material performs when both CO2 and water vapor are present simultaneously.
Sample and Measurement Conditions
A MOF-based adsorbent was selected for full characterization across all four measurement conditions. The measurement sequence proceeded from pore structure characterization through equilibrium isotherm analysis, TGA-based uptake testing under dry conditions at two CO2 concentrations, and finally dynamic breakthrough testing under humid conditions that represent the most realistic DAC scenario.
Measurement ApproachNitrogen adsorption and desorption isotherms at 77 K using the Matrix 1000, with BET surface area, micropore volume, and pore size distribution calculated from the dataCO2 adsorption isotherms at 298 K using the Matrix 1000, measured up to 100 kPaTGA CO2 uptake under pure CO2 at 25 degrees C following 80 degrees C pretreatmentTGA CO2 uptake under 400 ppm CO2 in N2 at 25 degrees C following 80 degrees C pretreatmentBreakthrough curve analysis using the BTsorb 100 at 400 ppm CO2, 40% relative humidity, 298 K, and 1 barThe nitrogen adsorption isotherm for the MOF material exhibited Type I behavior, which is characteristic of a predominantly microporous adsorbent with strong adsorbent-adsorbate interactions at low relative pressures. The key structural parameters measured from the nitrogen isotherm are:| Parameter | Value |
|---|---|
| BET Surface Area | 1160 m2/g |
| Micropore Volume | 0.438 cm3/g |
| Maximum Pore Width | 0.390 nm |
The maximum pore width of 0.390 nm confirms that the majority of the accessible pore network falls within the ultramicropore range, below 0.7 nm. This structural profile is well suited to CO2 adsorption at low pressures because the narrow pore geometry produces strong overlap of adsorption potential fields from opposing walls, enhancing the interaction energy with CO2 molecules relative to what would be observed in wider pores.Volumetric Isotherm at 298 K The CO2 adsorption isotherm collected at 298 K using the Matrix 1000 showed strong and continuous uptake with increasing pressure. At 298 K and 1 bar, the CO2 saturated adsorption capacity reached 119 cm3/g, equivalent to 5.431 mmol/g. This high capacity is consistent with the large BET surface area and high micropore volume measured from the nitrogen isotherm, and reflects the favorable interaction between CO2 molecules and the narrow ultramicropore environment. TGA Measurement Under Pure CO2 TGA measurements under pure CO2 at 25 degrees C yielded an uptake value of 4.42 mmol/g, in close agreement with the volumetric isotherm data. The consistency between these two independent measurement methods confirms the reliability of both the Matrix 1000 isotherm data and the TGA 1000 mass-based measurement.
TGA measurements under a 400 ppm CO2 in N2 mixture at 25 degrees C produced an uptake of 0.13 mmol/g, substantially lower than the 4.42 mmol/g measured under pure CO2. This reduction is a direct consequence of the relationship between CO2 partial pressure and adsorption behavior in ultramicropores. At 400 ppm, the CO2 partial pressure is approximately 0.04 kPa, far below the 1 bar (100 kPa) condition used in the pure-gas isotherm measurement. Lower partial pressure decreases the frequency of CO2-surface collisions and reduces the probability of pore filling, particularly in ultramicropores where adsorption is strongly pressure-dependent. This result illustrates an important distinction in DAC adsorbent evaluation: high saturated capacity under pure CO2 does not directly predict performance at ambient air CO2 concentrations. Both measurements are necessary, and the gap between them is a quantitative indicator of how steeply the adsorption isotherm falls off at low partial pressures.
Breakthrough testing using the BTsorb 100 at 400 ppm CO2 and 40% relative humidity produced a dynamic CO2 adsorption capacity of 0.004 mmol/g. This is significantly lower than the 0.13 mmol/g measured by TGA under dry 400 ppm conditions. The reduction in capacity under humid conditions is consistent with competitive adsorption by water vapor on hydrophilic surface sites within the MOF framework. At the low CO2 partial pressures relevant to DAC, water molecules present at 40% relative humidity can occupy adsorption sites more effectively than CO2, reducing the number of sites available for CO2 capture and limiting breakthrough capacity. This finding has direct implications for DAC adsorbent design. A material with strong CO2 uptake under dry conditions may not maintain that performance in real ambient air, where humidity levels vary and water vapor is always present. The breakthrough data quantifies this gap and identifies the specific operating condition, humid 400 ppm CO2, that represents the most stringent test of DAC-relevant performance. The results point toward specific material modification strategies: hydrophobic surface modification to reduce competitive water adsorption, amine functionalization to introduce CO2-selective binding sites that are less sensitive to humidity, or pore environment tuning to create an adsorption geometry that favors CO2 over water at low concentrations.
The four measurements conducted in this study together provide a complete picture of adsorbent behavior that no single method could deliver independently. The nitrogen physisorption data established the structural basis for adsorption performance, confirming high surface area and an ultramicropore-dominant pore size distribution. The CO2 isotherm at 298 K quantified maximum equilibrium capacity under idealized conditions. The TGA measurement at 400 ppm CO2 isolated the effect of low partial pressure under dry conditions. The breakthrough test at 400 ppm CO2 and 40% humidity added the competitive effect of water vapor, producing the most realistic assessment of DAC-relevant performance. Each step in this sequence narrows the gap between idealized laboratory measurement and real operating conditions. The progression from 5.431 mmol/g under pure CO2, to 0.13 mmol/g at 400 ppm dry, to 0.004 mmol/g at 400 ppm humid, quantifies both the partial pressure effect and the humidity effect independently, providing a clear diagnostic framework for understanding which factor limits performance and which material modifications are most likely to improve DAC applicability.
For research groups and development teams working on solid sorbents for DAC applications, the measurement workflow demonstrated here provides a structured approach to adsorbent evaluation that generates decision-relevant data at each stage. Pore structure data from the Matrix 1000 supports material comparison and synthesis optimization by linking structural parameters to adsorption potential. Equilibrium isotherm data establishes capacity benchmarks and provides input for thermodynamic modeling. TGA-based uptake measurements at dilute CO2 concentrations quantify performance at conditions relevant to ambient air without requiring a full dynamic experiment setup. Breakthrough testing with the BTsorb 100 under controlled humidity adds the final layer of realism needed to assess how candidate materials will behave in actual DAC operating environments. Together, these instruments provide a complete analytical pathway from initial material screening through realistic performance validation, reducing the number of iterations needed to identify materials worth advancing to larger-scale testing.
A multi-instrument characterization approach is most appropriate when:The adsorbent is being evaluated specifically for DAC or other low-concentration CO2 capture applications where ambient partial pressure is the relevant operating conditionMaterial development decisions depend on understanding both structural properties and performance under dilute and humid conditionsThe goal is to identify which performance limitation, partial pressure sensitivity or competitive water adsorption, is the primary barrier to improvementCandidate materials need to be ranked or compared on the basis of DAC-realistic performance metrics rather than idealized pure-gas capacityResearch findings will be used to guide surface modification, functionalization, or synthesis parameter changes aimed at improving humidity tolerance or low-pressure CO2 selectivityThis study demonstrates both the potential and the current limitations of a MOF-based adsorbent for direct air capture applications. The material possesses a well-defined ultramicropore structure with a BET surface area of 1160 m2/g and a micropore volume of 0.438 cm3/g, supporting a CO2 saturated capacity of 5.431 mmol/g under idealized conditions at 298 K and 1 bar. Performance decreases substantially at conditions relevant to actual DAC operation. TGA measurements at 400 ppm CO2 under dry conditions yielded 0.13 mmol/g, and breakthrough testing at 400 ppm CO2 with 40% relative humidity produced a dynamic capacity of 0.004 mmol/g. These results identify partial pressure sensitivity and competitive water adsorption as the two primary barriers to DAC applicability for this material class, and point directly toward surface hydrophobization and amine functionalization as modification strategies worth pursuing. From a measurement perspective, the combination of the AMI Matrix 1000, TGA 1000, and BTsorb 100 provided the analytical coverage needed to characterize each dimension of adsorbent performance independently and progressively. For research teams developing solid sorbents for DAC, this multi-instrument workflow offers a structured and complete path from structural characterization through realistic operational validation.
(1) McQueen, N.; Gomes, K. V.; McCormick, C.; Blumanthal, K.; Pisciotta, M.; Wilcox, J. A review of direct air capture (DAC): Scaling up commercial technologies and innovating for the future. Prog. Energy, 2021, 3, 032001. (2) Chuah, C. Y.; Ho, Y. L.; Syed, A. M. H.; Thivyalakshmi, K. G. K.; Yang, E.; Johari, K.; Yang, Y.; Poon, W. C. Applicability of adsorbents in direct air capture (DAC): Recent progress and future perspectives. Ind. Eng. Chem. Res. 2025, 64, 4117-4147. (3) Bose, S.; Sengupta, D.; Rayder, T. M.; Wang, X.; Kirlikovali, K. O.; Sekizkardes, A. K.; Islamoglu, T.; Farha, O. K. Challenges and opportunities: Metal-organic frameworks for direct air capture. Adv. Funct. Mater. 2024, 34, 2307478. (4) Sekizkardes, A. K.; Wang, P.; Hoffman, J.; Budhathoki, S.; Hopkinson, D. Amine-functionalized porous organic polymers for carbon dioxide capture. Mater. Adv. 2022, 3, 6668. (5) Bayati, B.; Keshavarz, F.; Rezaei, N.; Zendehboudi, S.; Barbiellini, B. New insight into impact of humidity on direct air capture performance by SIFSIX-3-Cu MOF. Phys. Chem. Chem. Phys. 2024, 26, 17645-17659.
Direct air capture removes CO2 from ambient air, where CO2 is present at approximately 400 parts per million and a partial pressure of about 40 Pa. Conventional carbon capture from flue gas operates at CO2 partial pressures of approximately 12 kPa, which is roughly 300 times higher. The much lower driving force in DAC imposes stricter requirements on adsorbent capacity, selectivity, and kinetics.At lower CO2 partial pressure, fewer CO2 molecules are available to collide with and adsorb onto the surface per unit time. In ultramicropores, pore filling is strongly pressure-dependent, meaning that the high capacity observed at 1 bar drops substantially when partial pressure falls to DAC-relevant levels. The TGA measurement at 400 ppm directly quantifies this effect under dry conditions.Water vapor can adsorb onto hydrophilic surface sites within the adsorbent framework, competing with CO2 for the same adsorption locations. At the very low CO2 partial pressures found in ambient air, this competition is particularly significant because water vapor is present at much higher partial pressure than CO2 under typical atmospheric humidity conditions. The result is a reduction in the number of sites available for CO2 capture.A breakthrough curve is a plot of outlet gas concentration versus time during a dynamic adsorption experiment. A gas mixture is passed through a packed bed of adsorbent, and the concentration of the target component in the outlet is monitored. A strongly adsorbing component takes longer to appear at the outlet, producing a delayed breakthrough. The area between the breakthrough curve and the inlet concentration line is used to calculate dynamic adsorption capacity under the specific flow and composition conditions tested.Thermogravimetric analysis measures the mass of a sample as a function of time and temperature. For CO2 uptake measurements, the sample is pretreated under controlled conditions to remove previously adsorbed species, then exposed to a CO2-containing gas stream at a defined temperature and flow rate. The increase in sample mass during exposure corresponds directly to the quantity of CO2 adsorbed, expressed in mmol per gram of adsorbent.
Learn how BET and t-range selection affects carbon black surface area accuracy, with NSA and STSA data across 11 standard carbon black grades.

Learn how static and dynamic BET methods compare for measuring silicon nitride surface area, with precise results and throughput insights from AMI Instruments.

Compare N2, Ar, and CO2 adsorbates to improve the characterization of nanoporous materials. Learn how to achieve more accurate and complete micropore analysis.

Learn how pretreatment method, temperature, time, and storage affect amorphous silica surface area and pore volume in nitrogen physisorption.