See how the Matrix 1000 BET analysis instrument delivers 4 alumina samples in 28 minutes with RSD below 0.3% across all ports, at production scale.
Ionic liquids have been studied extensively for their chemical tunability, thermal stability, and near-zero vapor pressure. In principle, they offer precisely the selectivity and capacity that gas separation, liquid purification, and catalysis researchers are looking for. In practice, using neat ionic liquids at scale presents persistent problems: high viscosity makes mass transfer slow, recovery from reaction or separation systems is difficult, and the cost of ionic liquid losses over multiple cycles limits economic viability.

Supported ionic liquids (SILs) are ionic liquids immobilized on porous solid carriers, either by physical deposition through methods such as impregnation, or by chemical anchoring through covalent bonding or sol-gel synthesis routes. The result is a solid-like composite that retains the chemical selectivity of the ionic liquid while gaining the mass-transfer and mechanical advantages of a structured porous matrix.
Ionic liquids themselves are room-temperature molten salts composed entirely of ions, typically an organic cation paired with an inorganic or organic anion. Their key properties include:
When immobilized on a porous support, these properties are retained while the viscosity and recovery challenges of neat ionic liquids are mitigated. SILs have demonstrated utility in gas and liquid adsorption and separation, as catalysts and catalyst supports, and in aqueous-phase removal of heavy-metal ions.
For all of these applications, the support’s specific surface area (SSA) and pore architecture are critical parameters. High SSA and well-matched pore sizes disperse thin ionic liquid films, maximize accessible interfacial sites, and shorten diffusion paths. These structural factors directly determine adsorption capacity, selectivity, and kinetics. Conversely, excessive ionic liquid loading into narrow pores can block transport pathways and reduce uptake rather than improve it.
This is why quantifying SSA and pore size distribution (PSD) through N2 and CO2 physisorption is an essential step before comparing adsorption performance across different ionic liquid formulations or support materials.
The primary design challenge in supported ionic liquid development is that chemical functionality and structural properties must both be optimized, and changes to one frequently affect the other. This creates several specific measurement and interpretation problems that researchers and process developers encounter regularly.
IL loading and pore blockage: Increasing ionic liquid loading on a support initially raises the density of active sites. Beyond an optimal loading level, excess ionic liquid begins to block pore entrances and reduce accessible surface area. This decreases uptake rather than increasing it, and the transition happens at a loading level that depends on pore size distribution. Without SSA and PSD data at each loading level, it is not possible to determine whether a capacity plateau reflects site saturation or pore blockage.
Support selection across material classes: Common supports for SILs include silica, activated carbon, alumina, metal-organic frameworks, zeolites, and polymers. Each has a different pore architecture, surface chemistry, and SSA range. The optimal support for a given ionic liquid and application depends on matching pore dimensions to the size of the ionic liquid ions and the target molecule, but this matching requires quantitative pore size distribution data rather than qualitative comparison.
Distinguishing transport limitations from site effects: When a supported ionic liquid shows lower than expected adsorption capacity or slow kinetics, the cause may be insufficient active site density, pore blockage reducing transport, or mismatched pore dimensions restricting diffusion. N2 and CO2 physisorption data, specifically BET SSA, total pore volume, and PSD, are the measurements that distinguish between these causes and direct the appropriate corrective action.
Comparative studies across formulations: Any meaningful comparison of supported ionic liquids with different cation or anion structures, different support materials, or different loading levels requires that the structural characterization of all materials is performed with the same method under the same conditions. Without this, apparent performance differences between formulations may reflect structural differences that were not measured rather than chemical differences that were intended.
When an ionic liquid is deposited on a porous support, it is distributed as a thin film within the pore network. The thickness of this film and the fraction of the pore surface it covers determine how many active sites are presented to the fluid phase. A support with high SSA provides more surface for film formation per unit mass of material, allowing a given quantity of ionic liquid to be dispersed as a thinner, more accessible film rather than pooling into thicker deposits that bury internal active sites.
Well-matched pore sizes, meaning pores large enough to accommodate the ionic liquid ions without steric restriction while remaining small enough to provide a high surface-to-volume ratio, are required for this dispersion to occur uniformly rather than selectively in certain pore size ranges.
Pore volume and pore size distribution also control how quickly molecules from the fluid phase can reach active sites within the supported ionic liquid. Larger total pore volume provides more pathways through the material. A PSD concentrated in the mesopore range (2 to 50 nm) supports faster diffusion than one dominated by micropores below 2 nm, where restricted diffusion can become rate-limiting.
For gas separation applications, fast kinetics are necessary for efficient column operation. For liquid-phase applications such as heavy metal removal, kinetics determine contact time requirements and the practical design of batch or flow systems.
Published research confirms that the relationship between ionic liquid loading and performance is not monotonic. For TMGL supported on SiO2, SO2 adsorption capacity comparable to neat ionic liquid was achieved with improved selectivity, but excess loading reduced accessible surface area and lowered uptake. This result, reported by Zhang et al. (2009), illustrates that SSA measurement at each loading level is required to identify the optimal loading rather than assume that more ionic liquid always produces better performance.
TMGL (tetramethylguanidinium lactate) supported on porous silica particles demonstrates SO2 adsorption capacity comparable to neat ionic liquid but with improved CO2/SO2 selectivity. The high SSA of the silica support enables thin film deposition that keeps active sites accessible. At excess loading, however, measured SSA declines as ionic liquid fills and partially blocks the pore network, and SO2 uptake drops correspondingly. This example shows directly how SSA measurement tracks with adsorption capacity as a function of loading.
A layered composite with an ionic liquid core, a ZIF-8 interlayer, and an ionic liquid shell addresses the aggregation problem that limits performance in simple IL-on-support systems. The ZIF-8 interlayer prevents ionic liquid aggregation, preserving accessible interfacial area and maintaining stability over adsorption and desorption cycling. CO2 capacity and selectivity both improve relative to simpler IL configurations. The structural explanation lies in the pore architecture provided by the ZIF-8 interlayer, which is why PSD characterization is necessary to understand why this architecture outperforms simpler alternatives.
Graphene oxide-based hydrogels incorporating ionic liquid functionality provide hierarchical porosity, a pore size distribution spanning multiple length scales from micropores through macropores, that combines high surface area with accessible transport pathways. This hierarchical architecture supports high heavy metal adsorption capacity and good regeneration performance across multiple cycles. The pore structure data connecting SSA and PSD to capacity and regenerability is what validates the structural design principle.
The most directly supported example from published research involves a graphene oxide/dicationic ionic liquid composite (GO-DIL) prepared by Shang et al. (2021) for the removal of hexavalent chromium (Cr(VI)) from water. This case study illustrates the full connection between structural characterization, mechanistic interpretation, and adsorption performance.
Graphene oxide (GO) and GO-DIL were characterized by N2 adsorption at 77 K using the AMI Micro 200 static physisorption analyzer. BET specific surface area and pore volume were derived from the adsorption and desorption isotherms. Adsorption tests confirmed that pH = 3 yielded the highest Cr(VI) uptake, consistent with maximum electrostatic attraction between protonated sites on GO-DIL and anionic chromium species under acidic conditions.
Sample | BET SSA (m2/g) | Pore Volume (cm3/g) |
|---|---|---|
Graphene Oxide (GO) | 9.93 | 0.043 |
GO-DIL | 10.46 | 0.077 |
GO-DIL showed higher BET specific surface area (10.46 vs 9.93 m2/g) and substantially higher pore volume (0.077 vs 0.043 cm3/g) compared to unmodified graphene oxide. The pore volume increase is particularly significant: GO-DIL provides nearly 80 percent more pore volume than GO, indicating that the dicationic ionic liquid modification creates additional accessible porosity rather than blocking existing pores.
The textural gain in GO-DIL directly aligns with the higher Cr(VI) adsorption capacity observed relative to unmodified GO. Kinetic analysis and Langmuir isotherm fitting point to an electrostatically driven mechanism: protonated imidazolium sites (N+) on the GO-DIL surface attract anionic Cr(VI) species under acidic conditions. The increase in accessible surface area and pore volume translates directly into more available electrostatic binding sites per gram of adsorbent.
The materials were further characterized by scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS), and FTIR spectroscopy, and adsorption/desorption cycling was evaluated to confirm regenerability. The N2 physisorption data from the AMI Micro 200 provided the structural foundation for interpreting all subsequent performance comparisons between GO and GO-DIL.
For research groups developing supported ionic liquids for industrial applications, and for process engineers evaluating SIL materials for deployment in separation or remediation systems, the practical value of accurate SSA and PSD measurement is direct and quantifiable.
Faster formulation optimization: Without SSA and PSD data at each ionic liquid loading level and for each support candidate, optimization requires exhaustive performance testing across all combinations. With structural data, researchers can identify which combinations are worth testing based on whether the pore architecture can support adequate IL film dispersion and molecular transport. This narrows the experimental space before performance testing begins.
Mechanistic clarity: When a supported ionic liquid shows unexpected capacity or kinetics, SSA and PSD data identify whether the cause is structural (pore blockage, insufficient surface area, mismatched pore dimensions) or chemical (wrong ionic liquid structure, insufficient active site density, pH-dependent site availability). This distinction directs the appropriate corrective action and avoids iterating on the wrong variable.
Reproducible comparative studies: SIL development programs typically compare multiple candidate materials measured under different conditions or synthesized by different routes. Standardized N2 physisorption characterization of all candidates using the same instrument and protocol creates a consistent structural baseline that makes performance comparisons interpretable and publishable.
Support selection confidence: The wide range of SSA and pore architectures across silica, activated carbon, alumina, MOFs, zeolites, and polymer supports means that support selection is consequential. PSD data measured on candidate supports before IL loading identifies which supports are structurally compatible with the ionic liquid size and target molecule dimensions, reducing the risk of committing synthesis resources to a structurally incompatible system.
Characterizing supported ionic liquids requires an instrument platform capable of resolving subtle differences in SSA and pore structure that may reflect changes as small as a difference in ionic liquid loading level or support surface treatment. Small differences in surface area and pore volume, such as the 0.53 m2/g SSA increase and 0.034 cm3/g pore volume increase between GO and GO-DIL in the Shang et al. study, must be measured reliably enough to support mechanistic interpretation and performance correlation.
The AMI Micro 200 static physisorption analyzer is built for exactly this level of sensitivity. Its high-vacuum stability, precise low-pressure transducer control, and robust cryogenic and thermostatic operation enable accurate BET SSA, total pore volume, and PSD measurements from ultralow P/P0 values through near-saturation. The instrument supports N2 at 77 K, Ar at 87 K, and CO2 at 195 K and 273 K, providing the multi-gas capability needed to characterize both mesopore and ultramicropore populations in a single platform.
Two integrated degassing ports and two independent analysis ports allow simultaneous sample preparation and measurement, improving throughput for comparative studies without reducing data quality. Automated dose and evacuation routines with tight transducer linearity deliver the repeatability that multi-formulation comparative studies require, where data from different measurement sessions must be compared with confidence.
Seamless switching among probe gases connects structural measurements directly to adsorption capacity, selectivity, and kinetics observed in downstream performance tests. For supported ionic liquid development programs where the relationship between pore structure and application performance must be established reliably and efficiently, the AMI Micro 200 provides the measurement foundation that makes those connections quantifiable.
Supported ionic liquids bridge the chemical versatility of ionic liquids with the practical handling advantages of porous solid materials. They have demonstrated utility in gas and liquid separation, catalysis, and environmental remediation, but their performance in each of these applications is governed by the structural properties of the support as much as by the chemical properties of the ionic liquid.
Specific surface area and pore size distribution determine how effectively the ionic liquid film is dispersed, how many active sites are accessible to the fluid phase, and how quickly molecules can reach those sites. The case study on GO-DIL for Cr(VI) removal confirms this directly: the higher pore volume of GO-DIL (0.077 vs 0.043 cm3/g) and slightly higher SSA (10.46 vs 9.93 m2/g) relative to unmodified GO provide the structural basis for interpreting its superior adsorption performance.
For research teams developing supported ionic liquids and for engineers evaluating them for deployment, N2 and CO2 physisorption characterization using a precise, multi-gas capable instrument is not an optional supporting measurement. It is the foundation on which meaningful performance comparisons and formulation optimization decisions are built. The Micro 200 developed by AMI instruments provides the sensitivity, vacuum performance, and multi-probe capability needed to make that foundation reliable.
Supported ionic liquids are ionic liquids immobilized on porous solid carriers, either through physical deposition or chemical bonding. Neat ionic liquids are used in liquid form, which creates handling challenges including high viscosity, difficult separation from reaction products, and losses during recovery and reuse. Immobilizing the ionic liquid on a solid support converts it into a manageable composite that retains the chemical selectivity of the ionic liquid while gaining the mechanical stability and mass-transfer characteristics of a porous solid.
For an ionic liquid supported on a porous material, the SSA determines how thinly the ionic liquid can be dispersed as a film within the pore network. Higher SSA allows the same mass of ionic liquid to be distributed across more surface area, producing a thinner film with more accessible active sites per gram of material. If SSA is too low, the ionic liquid forms thicker deposits that bury internal active sites and limit the accessible interfacial area available for adsorption or catalysis.
Pore size distribution describes how the internal pore volume of a material is distributed across different pore diameters. For supported ionic liquids, pore dimensions must be large enough to accommodate the ionic liquid ions without restriction and to allow target molecules to diffuse to active sites. A PSD concentrated in the mesopore range supports faster molecular transport than one dominated by micropores. Hierarchical PSDs spanning multiple size ranges can provide both high surface area and accessible transport pathways simultaneously.
Beyond an optimal loading level, excess ionic liquid begins to block pore entrances and fill transport pathways, reducing the accessible surface area and the rate of molecular diffusion through the material. This causes adsorption capacity and kinetics to decline even as the total mass of ionic liquid in the material increases. SSA and PSD measurements at each loading level identify where this transition occurs and define the optimal loading for a given support and ionic liquid combination.
GO-DIL is a graphene oxide/dicationic ionic liquid composite developed by Shang et al. for Cr(VI) removal from water. BET surface area measurements using the AMI Micro 200 showed that GO-DIL had a higher SSA (10.46 vs 9.93 m2/g) and substantially higher pore volume (0.077 vs 0.043 cm3/g) compared to unmodified graphene oxide. The higher pore volume indicates that the dicationic ionic liquid modification created additional accessible porosity. This textural improvement correlated with higher Cr(VI) adsorption capacity, attributed to increased availability of protonated imidazolium sites that attract anionic chromium species under acidic conditions.
See how the Matrix 1000 BET analysis instrument delivers 4 alumina samples in 28 minutes with RSD below 0.3% across all ports, at production scale.

Learn how silver powder specific surface area controls photovoltaic silver paste conductivity, sintering behavior, and solar cell efficiency across four materials.
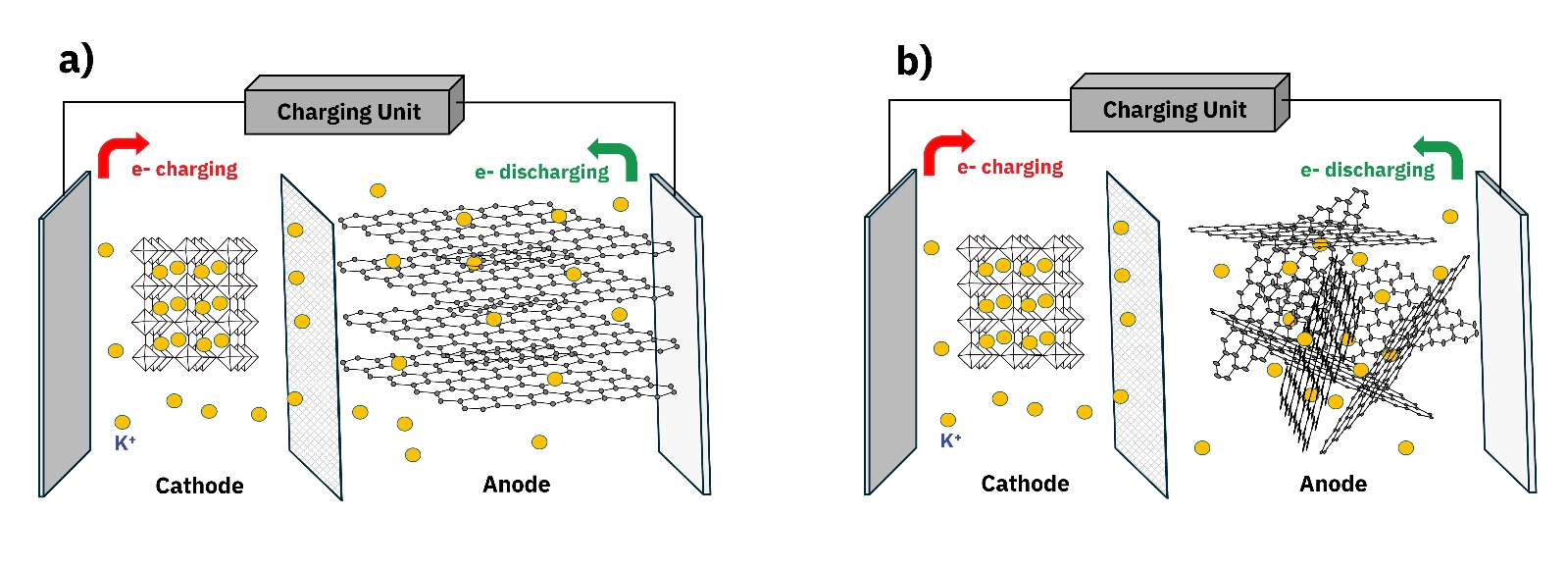
Discover how hard carbon anode surface area and pore structure drive potassium ion battery capacity, cycling stability, and Coulombic efficiency.

Learn how BET and t-range selection affects carbon black surface area accuracy, with NSA and STSA data across 11 standard carbon black grades.