Learn how specific surface area and pore structure control supported ionic liquid performance in adsorption, catalysis, and heavy metal removal applications.
Discover how hard carbon anode surface area and pore structure drive potassium ion battery capacity, cycling stability, and Coulombic efficiency.
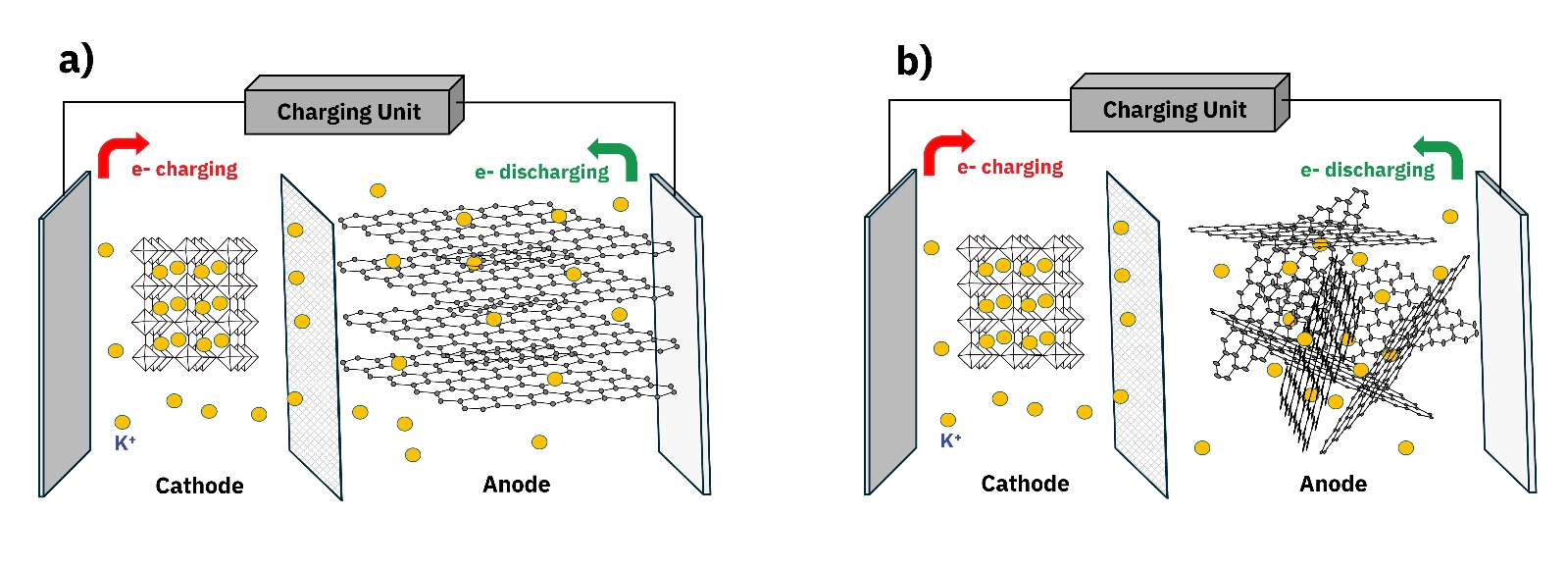
The race to develop viable alternatives to lithium-ion batteries is no longer theoretical. Potassium-ion batteries (PIBs) have moved to the center of that conversation, driven by the abundance of potassium raw materials, lower production costs, and a growing body of evidence that carbon-based anodes can deliver the cycling stability that practical applications require. But the gap between a carbon material that looks promising on paper and one that performs reliably in a battery cell often comes down to a single question: does the pore structure actually support potassium ion storage at the level the application demands? By using gas adsorption data collected on six hard carbon samples across two measurement methods, it shows how specific surface area and micropore volume vary significantly between materials that appear structurally similar, and why CO2 adsorption at 273 K provides critical information about ultramicropores that nitrogen adsorption at 77 K cannot access.
A hard carbon anode is a disordered, amorphous carbon material used as the negative electrode in potassium-ion batteries. Unlike graphite, which has a regular layered structure, hard carbon consists of randomly oriented sp2 carbon planes. This disordered arrangement produces inherently larger interlayer spacing, which facilitates faster intercalation and deintercalation of potassium ions (K+) without the structural modifications that graphite requires. Graphite has been studied extensively for PIBs because of its theoretical K+ storage capacity, but its practical performance is limited by the large ionic radius of K+, which restricts ion mobility through graphite channels. Attempts to expand graphite's interlayer spacing through surface modification often introduce structural defects that reduce performance and stability. Hard carbon avoids this problem by design, and when paired with an appropriate electrolyte, it has demonstrated excellent cycling stability. Within hard carbon, pore structure plays two distinct roles in battery performance:
Specific surface area governs the reaction efficiency between K+ ions in the electrolyte and the electrode surface. Higher surface area accelerates solid electrolyte interphase (SEI) film formation on the anode, which helps minimize electrolyte ion loss during early cycling.Pore volume determines how much K+ ion storage capacity is available within the pore channels of the anode material. Larger pore volumes, particularly in the mesopore range, have been shown to increase adsorption capacity and improve cycling stability.Both parameters must be characterized accurately to predict how a hard carbon material will perform in a real battery cell.Standard nitrogen adsorption at 77 K is the most widely used method for BET surface area and pore size distribution analysis. For many material classes, it provides sufficient information. For hard carbon anodes, it has a specific limitation that matters directly for potassium ion battery research. Nitrogen has a kinetic diameter of 0.364 nm. At 77 K, diffusion into pores narrower than approximately 1 nm is kinetically restricted, meaning the measurement cannot fully resolve the ultramicropore population in hard carbon materials. This is significant because hard carbon contains a meaningful fraction of pores in this sub-nanometer range, and these ultramicropores contribute to K+ ion storage capacity in actual battery operation. The consequence is that a laboratory relying solely on N2 adsorption at 77 K will undercharacterize the pore structure of hard carbon anodes and may draw incorrect conclusions about differences between materials. Two samples that appear to have similar pore structures from N2 data alone can show substantially different ultramicropore populations when measured with an appropriate probe. CO2 adsorption at 273 K resolves this limitation. CO2 has a smaller kinetic diameter of 0.33 nm, and at 273 K the saturated vapor pressure of CO2 is approximately 3.5 MPa, bringing the pressure range for micropore analysis into a moderate and accessible window of approximately 0.1 to 100 kPa. Gas diffusion at this temperature is fast, enabling access to pores smaller than 0.4 nm and producing high-resolution micropore distribution profiles that N2 cannot provide.
Before presenting the characterization data, it is worth establishing clearly why these structural measurements connect to electrochemical performance metrics that battery researchers and engineers care about. Solid Electrolyte Interphase Formation The SEI film forms at the electrode-electrolyte interface during initial charge-discharge cycles. It is a critical layer that allows K+ ions to pass while blocking electron transfer, and its quality directly affects first-cycle Coulombic efficiency and long-term capacity retention. A larger electrode surface area provides more sites for SEI formation and accelerates the process, helping the cell reach stable cycling behavior more quickly and with less irreversible capacity loss. K+ Ion Storage Capacity Potassium ions stored within the pore channels of the anode material contribute directly to charge-discharge capacity. Mesopore volume is particularly relevant here: published research by Quan et al. found that larger mesopore volumes increased adsorption capacity and improved cycling stability in porous carbon materials for PIBs. This finding establishes pore volume not just as a structural descriptor but as a predictor of practical battery capacity. Coulombic Efficiency and Battery Lifespan Changes in micropore size and volume in hard carbon anodes are closely related to the total K+ ion embedding capacity at the anode surface in actual battery systems. Superior pore structures, particularly those with well-developed ultramicropore networks, positively affect Coulombic efficiency, charge-discharge efficiency, and battery lifespan across cycling.
Materials
Six hard carbon samples were characterized, sourced from both commercial manufacturers and university research groups:Hard Carbon A and B: commercial samples characterized using N2 adsorption at 77 KHard Carbon C and D: university research samples characterized using CO2 adsorption at 273 KHard Carbon E and F: samples characterized using both N2 at 77 K and CO2 at 273 K for direct method comparisonMeasurement Conditions
N2 adsorption and desorption isotherms at 77 K, with BET surface area and differential pore size distributions calculated from the dataCO2 adsorption and desorption isotherms at 273 K, targeting ultramicropore characterization below 1 nmAll measurements conducted using the AMI Micro 200 physisorption analyzerNitrogen adsorption isotherms at 77 K revealed substantial differences in microporous character between the two commercial hard carbon samples.Hard Carbon B exhibited more pronounced microporous characteristics than Hard Carbon A, with a steeper uptake at low relative pressures indicating a denser ultramicropore population. The structural parameters calculated from the N2 data are compared below.
| Parameter | Hard Carbon A | Hard Carbon B |
|---|---|---|
| Specific Surface Area (m2/g) | 2.953 | 39.515 |
| Total Pore Volume (cm3/g) | 0.0087 | 0.041 |
| Micropore Volume (cm3/g) | not detected | 0.016 |
The surface area of Hard Carbon B (39.515 m2/g) was approximately 13 times larger than Hard Carbon A (2.953 m2/g). Hard Carbon B also showed a higher concentration of pores with average diameters below 1 nm in the differential pore size distribution. Both the total pore volume and the detected micropore volume of Hard Carbon B significantly exceeded those of Hard Carbon A. These differences in surface area and pore volume would be expected to produce meaningfully different battery performance, with Hard Carbon B providing more K+ ion storage sites and a larger electrode surface for SEI film formation.
CO2 adsorption at 273 K was used to characterize two university-prepared hard carbon samples, targeting their ultramicropore populations below 1 nm.| Parameter | Hard Carbon C | Hard Carbon D |
|---|---|---|
| CO2-probed Surface Area (m2/g) | 137.1 | significantly lower |
| Micropore Size (nm) | approximately 1.368 | not specified |
| Pore Volume (cm3/g) | 0.113 | significantly lower |
Hard Carbon C showed a CO2-probed specific surface area of 137.1 m2/g, a micropore size of approximately 1.368 nm, and a pore volume of 0.113 cm3/g, all significantly exceeding the corresponding values for Hard Carbon D. The CO2 isotherms and differential pore size distributions confirmed that Hard Carbon C contains a substantially more developed ultramicropore network, which would be expected to support higher K+ ion storage capacity.The direct comparison of N2 at 77 K and CO2 at 273 K on Hard Carbon E and F produced the most analytically instructive findings of the study. Both gases produced similar overall adsorption trends and isotherm shapes for the two samples. However, the two methods differed in their ability to resolve fine pore structure:
CO2 at 273 K characterized smaller-sized pores more effectively at low P/P0 values, accessing pores below 0.4 nm that N2 could not reach due to kinetic limitations at 77 KFor pore size and volume analysis in the sub-nanometer range, CO2 provided greater precision in the differential pore size distribution plotsN2 at 77 K underresolved the ultramicropore population, producing pore size distributions that missed features clearly visible in the CO2 dataThis comparison confirms that for hard carbon anodes, where the ultramicropore population is closely related to K+ ion embedding capacity, CO2 adsorption at 273 K is a necessary complement to N2 adsorption rather than an optional additional measurement.For research teams developing hard carbon anode materials for potassium-ion batteries, the measurement choices made during material screening determine the quality of the decisions that follow. If surface area and pore volume are measured using N2 only, the ultramicropore contribution to K+ storage capacity is systematically undercharacterized. This can lead to incorrect rankings of candidate materials, where samples with superior ultramicropore development are evaluated as similar to inferior candidates because the measurement method could not distinguish them. Resources committed to scaling up or optimizing the wrong material represent a direct and avoidable cost. For battery manufacturers evaluating commercially sourced hard carbon materials for anode qualification, the 13-fold difference in surface area between Hard Carbon A and Hard Carbon B in this study illustrates how significantly commercial materials can differ even within the same product category. Incoming material qualification based on N2-only BET data may not capture the ultramicropore differences that determine which material will perform better in the cell. Combining N2 at 77 K for broad surface area and mesopore characterization with CO2 at 273 K for ultramicropore analysis provides the complete structural picture needed to make reliable material selection and development decisions.
Researchers and development teams working on potassium ion battery anode materials require an instrument platform that can handle both N2 and CO2 adsorption measurements with the sensitivity and precision needed to resolve the pore structure differences that determine battery performance. The AMI Micro 200 physisorption analyzer is purpose-built for exactly this type of demanding characterization. It covers pore sizes from 0.35 nm to 500 nm, giving it the range to resolve ultramicropores that are critical for hard carbon anode analysis alongside larger pore features. Its turbo molecular pump supports high-vacuum operation, reducing residual gas interference at the low relative pressures where ultramicropore filling occurs. Two integrated degassing ports and two independent analysis ports allow simultaneous sample preparation and measurement, supporting higher throughput without compromising data quality. Reduced internal dead volume minimizes blank correction errors that can distort results in low-uptake materials like hard carbon, where precise pore volume measurement is essential. For battery research applications where the relationship between pore structure and electrochemical performance must be established reliably across multiple candidate materials, the AMI Micro 200 provides the measurement platform that makes that relationship quantifiable. It is the instrument used throughout this study to generate the surface area and pore structure data presented here, and it is equally suited to routine material qualification and advanced anode development programs.
Potassium ion battery performance at the anode is governed by structural properties that gas adsorption characterization can measure directly and reliably. The surface area and pore volume data presented here across six hard carbon samples show that significant structural differences exist between commercially and academically sourced materials, and that these differences are large enough to predict meaningful differences in K+ ion storage capacity, SEI film formation efficiency, and cycling stability. The comparison of N2 at 77 K and CO2 at 273 K demonstrates clearly that ultramicropore characterization in hard carbon anodes requires both methods. N2 alone underresolves the sub-nanometer pore population that contributes most directly to K+ ion embedding capacity. CO2 at 273 K fills that gap and provides the high-resolution pore size distributions needed to distinguish between candidate materials that N2 data cannot separate. For research programs and battery manufacturers that depend on accurate anode characterization to guide material development and qualification decisions, investing in the right measurement platform and the right measurement protocol is not a peripheral concern. AMI Instruments provides the precision and multi-method versatility needed to generate this data reliably. It builds a strong foundation for confident decisions on which materials to advance.
(1) Yang, K.; Zhou, W.; Fu, Q.; Xiao, L.; Mo, Y.; Ke, J.; Shen, W.; Wang, Z.; Tu, J.; Chen, S.; Gao, P.; Liu, J. Optimizing kinetics for enhanced potassium-ion storage in carbon-based anodes. Adv. Funct. Mater. 2023, 33, 2306190. (2) Li, X.; Li, J.; Ma, L.; Yu, C.; Ji, Z.; Pan, L.; Mai, W. Graphite anode for potassium ion batteries: Current status and perspective. Energy Environ. Mater. 2022, 5, 458-469. (3) Peng, Y.; Zhou, W.; Wang, Z.; Liu, H.; Fu, Q.; Xiao, K.; Wu, J.; Wang, Z.; Tu, J.; Gao, P.; Liu, J. Regulating anion chemistry with F-containing bonds enable superior potassium ions storage in hard carbon. Energy Storage Mater. 2023, 62, 1029. (4) Li, W.; Zhang, R.; Chen, Z.; Fan, B.; Xiao, K.; Liu, H.; Gao, P.; Wu, J.; Tu, C.; Liu, J. Microstructure- dependent K+ storage in porous hard carbon. Small, 2021, 17, 2100397. (5) Qian, Y.; Jiang, S.; Li, Y.; Yi, Z.; Zhou, J.; Tian, J.; Lin, N.; Qian, Y. Understanding mesopore volume- enhanced extra-capacity: Optimizing mesoporous carbon for high-rate and long-life potassium
Potassium is significantly more abundant and less expensive than lithium, and it does not require cobalt in the cathode chemistry. These factors make PIBs more cost-accessible and reduce supply chain risks associated with lithium and cobalt sourcing. Carbon-based anodes for PIBs can also leverage established manufacturing processes from lithium-ion battery production.Graphite's layered structure restricts K+ ion movement because the potassium ion has a larger ionic radius than lithium. Expanding graphite's interlayer spacing through modification often introduces defects that reduce performance and stability. Hard carbon's disordered structure provides inherently larger interlayer spacing without modification, facilitating faster K+ intercalation and deintercalation and better cycling stability.Nitrogen at 77 K has kinetic limitations that restrict adsorption into pores smaller than approximately 1 nm. Hard carbon contains a significant ultramicropore population in this sub-nanometer range that contributes to K+ ion storage capacity. CO2 at 273 K, with a smaller kinetic diameter of 0.33 nm and faster diffusion at the higher temperature, accesses these pores and provides high-resolution pore size distributions that N2 cannot deliver.Higher specific surface area increases the electrode-electrolyte contact area, which accelerates SEI film formation and improves reaction efficiency between K+ ions and the electrode. This leads to faster initial cycling stabilization and reduced irreversible capacity loss. Larger pore volumes additionally provide more physical space for K+ ion storage within the anode, contributing to higher charge-discharge capacity.There is no single best gas adsorbate for all nanoporous materials. Nitrogen is commonly used for general surface area and pore analysis, argon is preferred for polar surfaces due to its neutral behavior, and carbon dioxide is ideal for analyzing ultramicropores below 1 nm. Combining these adsorbates delivers the most reliable and comprehensive results.

Learn how specific surface area and pore structure control supported ionic liquid performance in adsorption, catalysis, and heavy metal removal applications.
See how the Matrix 1000 BET analysis instrument delivers 4 alumina samples in 28 minutes with RSD below 0.3% across all ports, at production scale.

Learn how silver powder specific surface area controls photovoltaic silver paste conductivity, sintering behavior, and solar cell efficiency across four materials.

Learn how BET and t-range selection affects carbon black surface area accuracy, with NSA and STSA data across 11 standard carbon black grades.