Learn how the Densi 100 True Density Analyzer is used to measure the true density of ceramic fracturing proppants. Ensure the quality of your proppants with AMI.[…]
Discover how specific surface area and pore structure affect the performance of potassium-ion batteries. Learn how AMI’s instruments can advance your battery research.
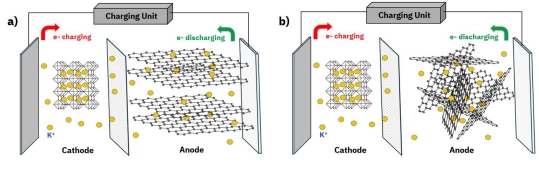
Potassium-ion batteries (PIBs) are considered an important complement to existing lithium-ion batteries (LIBs) due to their environmental friendliness, abundant raw material resources, and low cost. PIB anode materials have become the focus of research, and carbon-based anodes have been studied for their high conductivity and chemical stability. Graphite has been heavily studied for PIBs due to its theoretical storage capacity for K+ ions and therefore high energy density, shown schematically in Figure 1a. However, there are several limitations. The K+ ion has low mobility in graphite channels and pores due to its large ionic radius.⁽¹⁾ Many scholars have improved the performance of carbon materials in PIBs by surface modification, structural design, and functionalization.⁽¹˒²⁾ While graphite modification has been shown to improve K+ ion intercalation/deintercalation, attempts to increase the interlayer spacing often introduce defects into the material which reduce performance and stability. Alternatively, hard carbon is an amorphous material with randomly oriented sp2 planes. The disordered structure results in inherently larger spacing that facilitates faster intercalation and deintercalation of K+ ions. Because of this, hard carbon anodes have demonstrated excellent cycling stability when paired with an appropriate electrolyte.⁽³⁾ The optimization of cathodic material properties plays an integral role in PIB chemistry, and therefore surface area and pore size have received attention from scholars.⁽⁴⁾ Porous carbon materials have also emerged as promising anode materials for potassium-ion batteries due to their high specific surface area, excellent conductivity, and structural stability. Quan et al. prepared porous carbon materials with different pore structures, investigating the influence of pore volume on the performance of potassium-ion batteries. They found that larger mesopore volumes increased adsorption capacity and improved cycling stability.⁽⁵⁾

Figure 1: Working principle of potassium-ion batteries with a) graphite anode and b) hard carbon anode.
In this study, hard carbon materials were obtained from both a company and a university research group. Specific surface areas and pore structures were characterized with the AMI Micro 200 using N₂ and CO₂ adsorbate gases. The kinetic limitations of N₂ at 77K restrict adsorption in ultramicropores (< 1nm). The best solution is to use CO₂ (kinetic diameter: 0.33 nm) as the adsorbate at 273 K to analyze ultramicropores. At this ice point temperature, the saturated vapor pressure of CO₂ is very high (3.5 MPa), so the pressure required for micropore analysis falls within a moderate range (0.1 to 100 kPa). Additionally, at such high temperatures and relative pressures, gas diffusion is very fast, allowing access to pores smaller than 0.4 nm and yielding high-resolution micropore distribution profiles.
Large specific surface areas for potassium-ion battery anodes enhance the reaction efficiency between K+ ions in the electrolyte and the electrode, facilitating the faster formation of a solid electrolyte interphase (SEI) film on the anode. This helps to minimize electrolyte ion loss. Meanwhile, a larger pore volume provides higher capacity for potassium ion storage within the pore channels of the anode material, improving its charge-discharge capacity. From Figure 2, the adsorption-desorption curves obtained using N₂ at 77 K reveal that the Hard Carbon B sample exhibited more pronounced microporous characteristics compared to the Hard Carbon A sample, highlighted by a red oval in Figure 2a. Calculations show that the specific surface area of the Hard Carbon B sample (39.515 m²/g) was significantly larger than Hard Carbon A (2.953 m²/g). The differential pore size distribution plots are pictured in Figure 2b, which shows that Hard Carbon B had a higher number of pores with an average diameter below 1nm (ultramicropores). The total pore volume and micropore volume of Hard Carbon B (0.041 cm³/g and 0.016 cm³/g, respectively) were also greater than the total pore volume of Hard Carbon A (0.0087 cm³/g).
Figure 2: a) N₂ adsorption-desorption isotherm at 77K and b) differential pore size distribution plots for Hard Carbon A (black line) and Hard Carbon B (red line).
Figure 3: a) CO₂ adsorption-desorption isotherm at 273K and b) differential pore size distribution plots for Hard Carbon C (black line) and Hard Carbon D (red line).
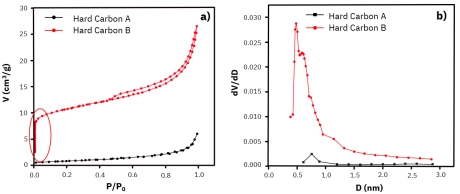
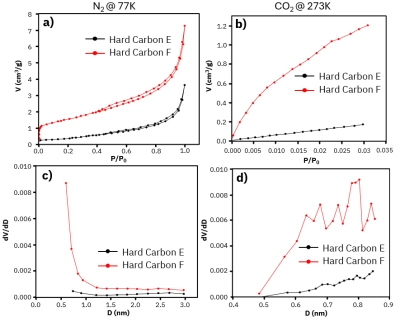
Figure 4: a) N₂ adsorption-desorption isotherm at 77K, b) CO₂ adsorption-desorption isotherm at 273K, c) differential pore size distribution plots using N₂, and d) differential pore size distribution plots using CO₂ for Hard Carbon E (black line) and Hard Carbon F (red line).
The specific surface area and pore volume of hard carbon materials can also be analyzed through CO₂ gas adsorption. As shown in Figure 3, two types of hard carbon materials prepared by a university research group were tested. The CO₂-probed specific surface area of Hard Carbon C reached 137.1 m²/g, with a micropore size of approximately 1.368 nm and a pore volume of 0.113 cm³/g, significantly surpassing the specific surface area and pore volume of Hard Carbon D. At a temperature of 273 K, the CO₂ pressure required for micropore size analysis falls within a moderate range (~0.1 to 100 kPa), and the diffusion rate of CO₂ is faster than N₂. Figure 4 compares the adsorption isotherms using N₂ and CO₂ for Hard Carbon E and F. It is evident that while the adsorption capacity and behavior trends were similar for both gases, CO₂ at 273 K was able to characterize smaller-sized pores more effectively at low P/P₀ values (Figure 4b). For pore size and volume analysis, CO₂ provided greater precision (Figure 4d). The changes in pore size and volume brought about by micropores are closely related to the total embedding capacity of K+ ions in the anode adsorption on the anode surface in actual battery systems. Superior pore structures positively impact Coulombic efficiency, charge-discharge efficiency, and battery lifespan.
In this application note, gas adsorption experiments using the AMI Micro 200, shown in Figure 5, demonstrated broad variations in the porosity of carbon battery materials. The use of N₂ adsorbate was compared to CO₂ adsorbate, and it was shown that CO₂ adsorbate was ideal for characterizing small micropores. The AMI Micro 200 is a high-precision instrument designed for the accurate determination of material properties important for battery research, such as specific surface area and pore size of electrodes.
For more information on our physisorption instruments, please contact us. You can also explore our full range of products or browse our technical library for more application notes.
(1) Yang, K.; Zhou, W.; Fu, Q.; Xiao, L.; Mo, Y.; Ke, J.; Shen, W.; Wang, Z.; Tu, J.; Chen, S.; Gao, P.; Liu, J. Optimizing kinetics for enhanced potassium-ion storage in carbon-based anodes. Adv. Funct. Mater. 2023, 33, 2306190.(2) Li, X.; Li, J.; Ma, L.; Yu, C.; Ji, Z.; Pan, L.; Mai, W. Graphite anode for potassium ion batteries: Current status and perspective. Energy Environ. Mater. 2022, 5, 458-469.(3) Peng, Y.; Zhou, W.; Wang, Z.; Liu, H.; Fu, Q.; Xiao, K.; Wu, J.; Wang, Z.; Tu, J.; Gao, P.; Liu, J. Regulating anion chemistry with F-containing bonds enable superior potassium ions storage in hard carbon. Energy Storage Mater. 2023, 62, 1029.(4) Li, W.; Zhang, R.; Chen, Z.; Fan, B.; Xiao, K.; Liu, H.; Gao, P.; Wu, J.; Tu, C.; Liu, J. Microstructure dependent K+ storage in porous hard carbon. Small, 2021, 17, 2100397.(5) Qian, Y.; Jiang, S.; Li, Y.; Yi, Z.; Zhou, J.; Tian, J.; Lin, N.; Qian, Y. Understanding mesopore volume enhanced extra-capacity: Optimizing mesoporous carbon for high-rate and long-life potassium storage. Energy Storage Mater. 2020, 29, 341-349.Specific surface area directly affects the interaction between K⁺ ions and the anode material. A larger surface area enhances ion adsorption, accelerates solid electrolyte interphase (SEI) formation, and improves reaction kinetics. This leads to better charge–discharge efficiency, higher Coulombic efficiency, and improved cycling stability in potassium-ion batteries (PIBs).
Pore structure determines how efficiently K⁺ ions can be stored and transported within the anode. Micropores (<2 nm) and ultramicropores (<1 nm) increase potassium ion adsorption capacity, while mesopores improve electrolyte diffusion. An optimized pore size distribution enhances storage capacity, rate performance, and battery lifespan.
Hard carbon offers a disordered structure with larger interlayer spacing compared to graphite, allowing easier intercalation and deintercalation of large K⁺ ions. This reduces kinetic limitations and improves cycling stability. In contrast, graphite may suffer from slower ion mobility and structural instability due to potassium’s larger ionic radius.
N₂ adsorption at 77 K has diffusion limitations in ultramicropores (<1 nm). CO₂ adsorption at 273 K overcomes this issue due to its smaller kinetic diameter and faster diffusion rate. As a result, CO₂ provides more accurate characterization of ultramicropores, which are critical for potassium-ion storage performance.
The AMI Micro 200 enables high-precision measurement of specific surface area, pore size distribution, and pore volume using N₂ and CO₂ adsorption techniques. These parameters are essential for optimizing hard carbon anodes and improving battery capacity, efficiency, and lifespan. The instrument provides reliable data to support advanced electrode material development.
Learn how the Densi 100 True Density Analyzer is used to measure the true density of ceramic fracturing proppants. Ensure the quality of your proppants with AMI.[…]
Learn about pulse chemisorption, a technique for characterizing catalysts and measuring metal surface area. Discover how AMI’s instruments can help you.[…]
Learn how differential scanning calorimetry (DSC) is used to analyze the thermal properties of lipstick. See how the AMI DSC 600 can help you develop high-quality cosmetics.[…]
Understanding TPD kinetics, surface characterization, and desorption dynamics for heterogeneous catalyst analysis.[…]