Learn how the Densi 100 True Density Analyzer is used to measure the true density of ceramic fracturing proppants. Ensure the quality of your proppants with AMI.[…]
Learn about pulse chemisorption, a technique for characterizing catalysts and measuring metal surface area. Discover how AMI's instruments can help you.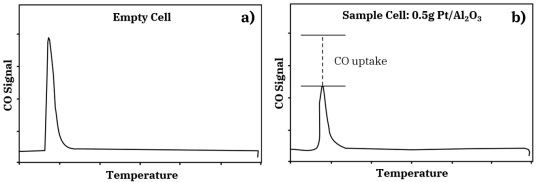
Previous issues of AMI Notes have discussed different selective chemisorption techniques and how they may be used to determine the specific metal surface area of supported metal catalysts. One additional technique commonly used for the same purpose is pulse chemisorption. This method is one of the simplest, most straightforward ways to measure adsorbate uptake by a metal surface; however, as with most other measurements in catalysis, interpretation of the results can be problematic if the nature of the catalyst system and the experiment itself are not well-understood. Pulse chemisorption is often used to calculate the particle dispersion and surface area of reduced metal catalysts supported on metal oxides.Both CO and H₂ are commonly used adsorbates; CO equilibrates quickly and adsorbs more strongly to most metals, while H₂ is effective and non-toxic. A 2025 study by Kanuri et al. used an AMI 300 Chemisorption Analyzer with H₂ adsorbate to calculate the dispersion and metallic surface area of Cu⁰ in CuO-ZnO-CeO₂ catalysts.⁽¹⁾ Combined with X-ray diffraction (XRD) and scanning electron microscopy (SEM), H₂ pulse chemisorption was used to determine which synthesis method yielded the highest Cu dispersion. However, adsorbate gas is not limited to CO and H₂. Warmuth et al. also used an AMI 300 with N₂O gas to calculate the surface area of Cu⁰ catalyst supported on ZnO/ZrO₂ and ZnO/ZrO₂/SiO₂.⁽²⁾ They were able to quantify the decrease in Cu⁰ surface area as a function of reaction time on stream.

Figure 1: Schematic comparing the pulse chemisorption process with an empty sample cell (top) and a sample cell with adsorbent material (bottom)This AMI Note discusses the principles of pulse chemisorption experiments as well as some common experimental observations.Pulse chemisorption measurements are usually carried out in an atmospheric pressure flow system at ambient temperature. A small catalyst sample (0.1-1.0 g) is placed in a small diameter cell usually of quartz or stainless-steel construction. After appropriate pretreatment to provide a clean, reduced metal surface, the sample is subjected to flow of an inert gas such as Ar. A small amount of chemisorbing gas is injected into this Ar stream before it flows over the catalyst. The composition of the stream exiting the sample cell is monitored quantitatively by a thermal conductivity detector (TCD) or similar device, and this signal is compared to that produced by the injected pulse of chemisorbing gas flowing through the cell in the absence of the sample. This process is shown schematically in Figure 1. If the amount of chemisorbing gas in the injected pulse is known, the uptake by the catalyst may be calculated, as shown in Figure 2. Often, very little signal is detected after the initial pulse of adsorbate over a clean catalyst surface. This indicates that the catalyst surface has adsorbed all of the chemisorbing gas in the pulse. Pulse 1 in the sequence shown in Figure 3a demonstrates this complete uptake. This pulse procedure may be repeated many times over the same sample. As the catalyst surface becomes saturated with adsorbate, less and less of the gas in each pulse is taken up by the sample. With each pulse, the size of the detector signal increases until it becomes constant and equal to the blank cell pulse signal. The total chemisorption uptake by the sample is then given by the sum of the uptakes from all of the pulses prior to saturation. The sequence of CO pulses shown in Figure 2 ends with saturation of the surface.
Figure 2: A 50 microliter pulse of CO in He a) after exiting an empty sample cell and b) after exiting a sample cell containing 0.5 g Pt/Al₂O₃; the difference in area of the two pulses gives the amount of CO taken up by the catalyst surfacePulse chemisorption experiments can usually be completed more rapidly than other selective chemisorption techniques used for determining metal surface areas for the following reasons: ✓ It entails the equilibrium adsorption of adsorbate under sub-atmospheric pressure, which can be completed in less than an hour. ✓ Pulse chemisorption requires fewer steps compared to temperature-programmed desorption (TPD). Pulse chemisorption experiments can also be standardized more readily than the other selective chemisorption techniques because there are fewer parameters to set (gas flow rate, sample temperature, and pulse size). Volumetric chemisorption and TPD experiments require more steps and therefore more interacting parameters. In practice, pulse chemisorption is often the preferred method of determining metal surface areas because of the advantages described above. However, some features of the pulse chemisorption technique make it difficult to use and can provide misleading results for some catalyst systems.
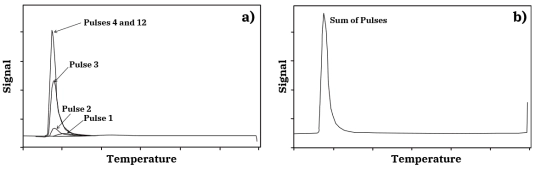
Figure 3: A sequence of pulses of CO in He over a clean Pt/Al₂O₃ catalyst resulting in complete saturation of the surfaceFlow techniques such as pulse chemisorption and TPD often give misleadingly lower uptakes than volumetric chemisorption due to the following: ✓ Inherent difficulty in measuring weakly held adsorbate molecules under inert gas flow, in which the gas desorbs in a slow, continuous manner which only adds to the baseline signal, and; ✓ Slow adsorption kinetics at the experiment temperature. Pulse chemisorption experiments have been carried out with a wide variety of catalysts however, and when used properly, the technique can become a routine and useful method of catalyst characterization.
Metal particle dispersion and surface area are important variables in catalyst development, often affecting the catalyst stability and rate of conversion. Pulse chemisorption is a straightforward and effective method for measuring these parameters, and AMI offers several user-friendly options for pulse chemisorption analysis, shown in Figure 4. The AMI 300 has proven to provide accurate and impactful pulse chemisorption results for catalytic research,(1-3) and the new AMI 400 boasts enhanced safety, high precision, and customizability at a competitive price point.
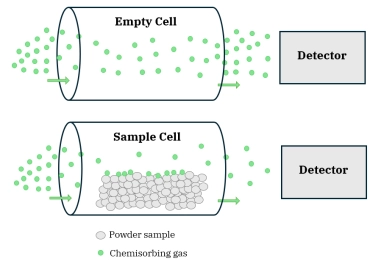
Figure 4: Highlight of the AMI 300 and AMI 400 Chemisorption AnalyzersFor more information on our chemisorption instruments, please contact us. You can also explore our full range of products or browse our technical library for more application notes.
(1) Kanuri. S.; Singh, S.A.; Dinda, S. Investigation of solvent and stirring-assisted morphologically tuned CuO-ZnO-CeO₂ catalyst for CO₂ hydrogenation to methanol. ACS Omega, 2025, 10, 62715-62726.(2) Warmuth, L.; Hofsäß, C. Zevaco, T. A.; Schild, D.; Pitter, S.; Sauer, J. Investigation of methanol catalyst stability in presence of potential green hydrogen impurities. Int. J. Hydrogen Energy, 2026, 207, 153382.
(3) Balderas, R. I.; Myers, L.; Miller, J.; Wrasman, C. J.; Vardon, D.; Richards, R. M. Bifunctionality of supported metal hydrodeoxygenation catalysts. RSC Sustainability, 2025, 3, 2984-3001.Pulse chemisorption is a catalyst characterization technique used to measure the uptake of a chemisorbing gas on a metal surface. It is commonly applied to determine metal dispersion and metallic surface area in supported metal catalysts. The method involves injecting controlled pulses of adsorbate gas over a pretreated catalyst and measuring the amount adsorbed.
Carbon monoxide (CO) and hydrogen (H₂) are the most frequently used adsorbates. CO adsorbs strongly and equilibrates quickly with many metal surfaces, while H₂ is non-toxic and widely applicable. In some systems, other gases such as N₂O may be used to selectively measure surface metal species, depending on the catalyst and research objective.
Metal dispersion is calculated by measuring the total amount of chemisorbing gas adsorbed by the catalyst surface before saturation. By knowing the stoichiometric relationship between the adsorbate molecules and surface metal atoms, researchers can determine the number of exposed metal atoms and calculate dispersion and active surface area.
Pulse chemisorption is faster and simpler than many other selective chemisorption methods, such as volumetric chemisorption or temperature-programmed desorption (TPD). It requires fewer adjustable parameters, can often be completed in less than an hour, and is relatively easy to standardize, making it a preferred routine technique in many catalyst research laboratories.
Pulse chemisorption may underestimate adsorption in systems with weakly bound adsorbates or slow adsorption kinetics. Under continuous inert gas flow, weakly adsorbed species may desorb gradually and contribute to baseline signals, potentially leading to lower measured uptakes. Proper experimental design and understanding of the catalyst system are essential for accurate interpretation.
Learn how the Densi 100 True Density Analyzer is used to measure the true density of ceramic fracturing proppants. Ensure the quality of your proppants with AMI.[…]
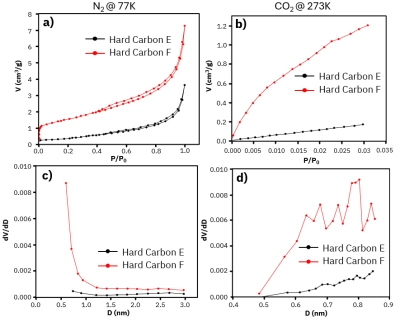
Discover how specific surface area and pore structure affect the performance of potassium-ion batteries. Learn how AMI’s instruments can advance your battery research.[…]
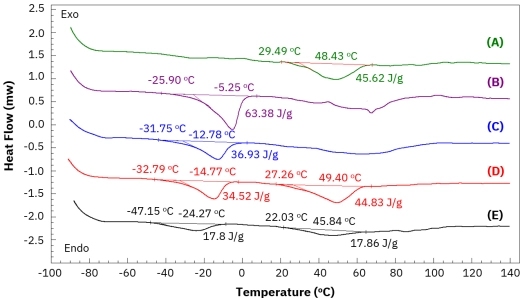
Learn how differential scanning calorimetry (DSC) is used to analyze the thermal properties of lipstick. See how the AMI DSC 600 can help you develop high-quality cosmetics.[…]
Understanding TPD kinetics, surface characterization, and desorption dynamics for heterogeneous catalyst analysis.[…]